Research Article
Volume 3 Issue 1 - 2018
Antipyretic Effect of Methanolic Extracts of Limnophila repens and Argyreia cymosa Whole Plant on Albino Rats
1Department of Pharmacognosy and Phytochemistry, A. M. Reddy Memorial College of Pharmacy, India
2Department of Pharmacognosy and Phytochemistry, Gitam University, India
2Department of Pharmacognosy and Phytochemistry, Gitam University, India
*Corresponding Author: Venkateswarlu Gunji, Department of Pharmacognosy and Phytochemistry, A. M. Reddy Memorial College of Pharmacy, India.
Received: October 13, 2018; Published: October 31, 2018
Abstract
Objective: The aim of the study was to evaluate the antipyretic activity of methanolic whole plant extracts of Limnophila repens (L. repens) and Argyreia cymosa (A. cymosa) using Brewer’s yeast-induced pyrexia model.
Materials and Methods: It was a randomized controlled experimental study. A total of 60 rats were taken, dividing them into six groups, each containing ten rats. Methanolic extract of L. repens (MELR) and A. cymosa (MEAC) was administered at 200, and 400 mg/kg doses orally to the respective four groups. The control group was fed with normal saline at 2 ml/kg. A 20% suspension of Brewer's yeast in normal saline was injected subcutaneously at a dose of 10 ml/kg body weight under the nape of the throat of rodents in all groups. Pyrexia produced following ten h of Brewer's yeast injection, and the temperature was recorded. Drugs received soon after the development of pyrexia and temperatures were recorded. Paracetamol at 150 ml/kg orally was taken as the conventional medication.
Results: The MELR and MEAC were showed significant (P<0.05) antipyretic activity at 400 mg/kg. Paracetamol showed substantial antipyretic activity from 30 min of drug administration to 180 min. At 400 mg/kg dosage the extracts (MELR and MEAC) revealed a considerable decrease in yeast evoked raised temperature when compared with that of standard drug paracetamol where by the extract dose 200 mg/kg had been less effective as compared to the higher dose (p <0.05).
Conclusion: This research confirmed that MELR and MEAC at a dose of 200 and 400 mg/kg own considerable antipyretic outcome against the yeast-induced raised temperature. However, the active chemical constituents responsible for the antipyretic action need to be investigated further.
Keywords: Antipyretic; Paracetamol; Phytochemical screening; Limnophila repens and Argyreia pilosa
Introduction
For years, natural treatments and particularly therapeutic vegetation had been the primary or even the sole alternative of our ancestors for their remedy. However, despite the development of the pharmaceutical industry, medicinal plants and treatments that could be drawn were never abandoned entirely, and people continue to resort to traditional medicine [1]. Natural products are believed to be an essential source of new chemical substances with the potential therapeutic application. Several plant species were traditionally used as analgesics. In general, the herbal plant usage in the treatment of disease and pain relief is one of the important strategies in medicine [2].
The genus Limnophila is frequently used in the traditional medicine against cardiovascular diseases, stomach disorders, elephantiasis, diarrhoea, dyspepsia, fever, dysentery, indigestion, Dysmenorrhoea and abdominal pain [3-5], Phytochemical analysis of genus Limnophila revealed the presence of the number of phytoconstituents such as flavonoids, tannins, alkaloids, terpenoids, steroids, and glycosides [6]. This diversity in compounds could justify the traditional use of L. repens.
The genus Limnophila is relatively abundant and widely used in folk medicine as an antioxidant [7,8], antimicrobial [9] anticancer [10], antimycobacterial [11], as on to date no biological studies have been conducted on this plant.
Argyreia cymosa is a decorative, as well as a therapeutic herb. Every part of the herb will be traditionally used being a folklore medication for the management of numerous illnesses by the Indian traditional healer. Its root is employed to remedy numerous disease like STD viz., gonorrhea and syphilis, blood diseases. Conventionally, the paste of the leaves is placed on the throat area for coughing, quinsy and then used outwardly in case of itch, dermatitis and other skin troubles, antidiabetic, antiphlogistic, rheumatism and minimize burning sensation [12]. A vast range of phytochemical constituents has been separated from the genus Argyreia, i.e., glycosides, alkaloids, amino acids, proteins, flavonoids, triterpene and steroids [13]. The plant has been reported a few biological activities including Antibacterial [14] and antioxidant [14]. Even though the drug has many uses, it's pharmacological, and phytochemistry is very poorly explored.
Traditionally, both plants were utilized for antipyretic activity, but till date, no scientific evidence has been reported. Therefore, the current study has been carried out with the methanolic extract of L. repens (MELR) and A. cymosa (MEAC) to investigate its antipyretic activity against baker's yeast-induced fever in rabbits using paracetamol as a reference standard.
Materials and Methods
Plant Material
The plants L. repens and A. cymosa were collected from Tirupathi, Chittoor district of Andhra Pradesh was authenticated at Botany Division, Sri Venkateswara University. The voucher specimen (No. 1568 and 1043) were deposited at herbarium and raw drug depository respectively. The plant components had been dried out under shade for Two weeks, coarsely powdered and then kept in air restricted canisters guarded against humidity and sunlight for further study.
The plants L. repens and A. cymosa were collected from Tirupathi, Chittoor district of Andhra Pradesh was authenticated at Botany Division, Sri Venkateswara University. The voucher specimen (No. 1568 and 1043) were deposited at herbarium and raw drug depository respectively. The plant components had been dried out under shade for Two weeks, coarsely powdered and then kept in air restricted canisters guarded against humidity and sunlight for further study.
Preparation of Extract
About 250g of the powdered crude drug of L. repens and A. cymosa were extracted by cold maceration with 1000 mL of methanol for 18 h after pretreatment with petroleum ether. Both extracts obtained had been concentrated to dryness in the vacuum at 40oC and kept at 4oC inside the refrigerator till further used. The extracts had been subjected to phytochemical and pharmacological evaluation.
About 250g of the powdered crude drug of L. repens and A. cymosa were extracted by cold maceration with 1000 mL of methanol for 18 h after pretreatment with petroleum ether. Both extracts obtained had been concentrated to dryness in the vacuum at 40oC and kept at 4oC inside the refrigerator till further used. The extracts had been subjected to phytochemical and pharmacological evaluation.
Phytochemical Screening
The various extract of L. repens and A. cymosa were subjected to qualitative chemical analysis by using standard procedures [11-14].
The various extract of L. repens and A. cymosa were subjected to qualitative chemical analysis by using standard procedures [11-14].
Animal used
Adults Wistar albino rats of either sex weighing 180-200g each were used. The animals had been maintained in the regular metallic cages in sets of 6 per cage, with free access to standard diet and drinking water ad libitum in the animal house and kept at room temperature under suitable dietary and environmental circumstances throughout the experiment. The Institutional Animal Ethics Committee examined the complete animal protocols ahead of performing the trials.
Adults Wistar albino rats of either sex weighing 180-200g each were used. The animals had been maintained in the regular metallic cages in sets of 6 per cage, with free access to standard diet and drinking water ad libitum in the animal house and kept at room temperature under suitable dietary and environmental circumstances throughout the experiment. The Institutional Animal Ethics Committee examined the complete animal protocols ahead of performing the trials.
Drugs, reagents, and apparatus used
Paracetamol, Baker yeast, Distilled water and Digital Thermometer.
Paracetamol, Baker yeast, Distilled water and Digital Thermometer.
Acute toxicity study
To evaluate the degree of toxicity of L. repens methanolic extract, the acute toxicity study was worked based upon OECD (Organization for Economic Cooperation and Development) 423 recommendations to the dose of 2000 mg/Kg. The experimental animals had been noticed for 1h constantly after which hourly for 4h and lastly every 24h up to 14 days for any physical symptoms of the level of toxicity, including writhing, gasping, palpitations and lowered respiratory rate or mortality. No animals died. Therefore the LD50 is greater than 2000 mg/kg. Pre-screening investigation with 200 and 400mg per body weight was done [15].
To evaluate the degree of toxicity of L. repens methanolic extract, the acute toxicity study was worked based upon OECD (Organization for Economic Cooperation and Development) 423 recommendations to the dose of 2000 mg/Kg. The experimental animals had been noticed for 1h constantly after which hourly for 4h and lastly every 24h up to 14 days for any physical symptoms of the level of toxicity, including writhing, gasping, palpitations and lowered respiratory rate or mortality. No animals died. Therefore the LD50 is greater than 2000 mg/kg. Pre-screening investigation with 200 and 400mg per body weight was done [15].
Anti-pyretic activity
The animals were randomly divided into six groups, each group consisting of 10 rats; a total of 60 rats were used in the study by randomized sampling technique: group I (control, normal saline given orally at 2 ml/kg body weight); group II (standard, paracetamol 150 mg/kg); group’s III and IV [methanolic extract of L. repens (MELR)], V and VI [methanolic extract of A. cymosa (MEAC) 200, and 400 mg/kg, respectively]. This is certainly a traditional way of antipyresis screening. Wistar strain of albino rats of either sex weighing 100-200 g was used for the study. The animals had fasted for 18 h before the beginning of the test, yet water was supplied ad libitum. The initial rectal temperature was recorded by using a rectal thermometer to a depth of 1.5 cm in the rectum of rodent. Animals with a body temperature between 36 and 38°C had been as part of the research. A 20% Brewer's yeast in 0.9% w/v saline was injected subcutaneously under the nape of the throat at a dose of 10 ml/kg thereafter. The injection site was rubbed so that the spread of suspension under the skin. Room temperature was maintained at 22-24°C. Following the yeast injection, food was instantly withdrawn. After 10 h post-challenge, the rise in rectal temperature was recorded. Animals which demonstrated an increase in body's temperature to 39°C had been included in the research, enabling the minimum of six rats in every group, the total of 36 rats. The animals received the standard (paracetamol 150 mg/kg) or the test compound (MELR and MEAC 200 and 400 mg/kg) by oral administration, and the rectal temperature was recorded at 0, 30, 60, 90, 120 and 180 min after dosing. The maximum reduction in average rectal temperature in comparison with the control hyperpyrexia group was calculated and compared [16].
The animals were randomly divided into six groups, each group consisting of 10 rats; a total of 60 rats were used in the study by randomized sampling technique: group I (control, normal saline given orally at 2 ml/kg body weight); group II (standard, paracetamol 150 mg/kg); group’s III and IV [methanolic extract of L. repens (MELR)], V and VI [methanolic extract of A. cymosa (MEAC) 200, and 400 mg/kg, respectively]. This is certainly a traditional way of antipyresis screening. Wistar strain of albino rats of either sex weighing 100-200 g was used for the study. The animals had fasted for 18 h before the beginning of the test, yet water was supplied ad libitum. The initial rectal temperature was recorded by using a rectal thermometer to a depth of 1.5 cm in the rectum of rodent. Animals with a body temperature between 36 and 38°C had been as part of the research. A 20% Brewer's yeast in 0.9% w/v saline was injected subcutaneously under the nape of the throat at a dose of 10 ml/kg thereafter. The injection site was rubbed so that the spread of suspension under the skin. Room temperature was maintained at 22-24°C. Following the yeast injection, food was instantly withdrawn. After 10 h post-challenge, the rise in rectal temperature was recorded. Animals which demonstrated an increase in body's temperature to 39°C had been included in the research, enabling the minimum of six rats in every group, the total of 36 rats. The animals received the standard (paracetamol 150 mg/kg) or the test compound (MELR and MEAC 200 and 400 mg/kg) by oral administration, and the rectal temperature was recorded at 0, 30, 60, 90, 120 and 180 min after dosing. The maximum reduction in average rectal temperature in comparison with the control hyperpyrexia group was calculated and compared [16].
Statistical Analysis
Statistical analysis was carried out using Graph Pad Prism 5.0 (Graph Pad Software, San Diego, CA). All results were expressed as mean ± SD. The data were analyzed by one-way ANOVA followed by Tukey multiple comparison tests.
Statistical analysis was carried out using Graph Pad Prism 5.0 (Graph Pad Software, San Diego, CA). All results were expressed as mean ± SD. The data were analyzed by one-way ANOVA followed by Tukey multiple comparison tests.
Results
Acute Toxicity Studies
The MELR and MEAC, when orally administered in the dose of 2000 mg/kg body wt. did not produce any significant changes in the autonomic or behavioural responses, including death during the observation period.
The MELR and MEAC, when orally administered in the dose of 2000 mg/kg body wt. did not produce any significant changes in the autonomic or behavioural responses, including death during the observation period.
Phytochemical Screening
The phytochemical screening for methanolic extracts of L. repens and A. cymosa were carried out, and results were displayed in Table 1.
The phytochemical screening for methanolic extracts of L. repens and A. cymosa were carried out, and results were displayed in Table 1.
| Phytoconstituents | Method | MEAC | MELR |
| Flavonoids | Shinoda Test | + | + |
| Zn. Hydrochloride test | + | + | |
| Lead acetate Test | + | + | |
| Volatile oil | Stain test | - | - |
| Alkaloids | Wagner Test | + | + |
| Hager’s Test | + | + | |
| Tannins & Phenols | Fecl3 Test | + | + |
| Potassium dichromate test | + | + | |
| Saponins | Foaming Test | - | - |
| Steroids | Salkowski test | + | + |
| Carbohydrates | Molish test | - | + |
| Acid compounds | Litmus test | + | - |
| Glycoside | Keller-Killani Test | + | + |
| Amino acids | Ninhydrin test | + | + |
| Proteins | Biuret | + | + |
Table 1: Phytochemical analysis of the methanolic extract of Argyreia cymosa (MEAC) and methanolic extract of Limnophila repens (MELR).
“+” – Present and “-” - Absent
“+” – Present and “-” - Absent
Antipyretic Activity
Subcutaneous injection of the pyrogenic dose of yeast produced elevated changes in rectal temperature, which is shown in Table 2. The MELR and MEAC showed a significant (P<0.05) decrease in rectal temperature at dose 400 mg/kg when compared with the standard. Extract at doses of 200, and 400 mg/kg showed a progressive decline in mean temperature pattern with the increase in the dose. Paracetamol showed significant (P<0.001) decrease in rectal temperature. The onset of action of paracetamol was 30 min. The line graph (Fig. 1) shows that paracetamol registered a phenomenal decrease in mean temperature from 38.7±0.12 to 37.01±0.42. Figure 2 illustrates that the most significant (P < 0.05) decrease in round-the-clock mean temperature in this study was shown by paracetamol followed by MELR and MEAC at 400 mg/kg and 200 mg/kg. Statistical analysis was performed using one-way analysis of variance followed by Tukey's test. The consequences of extract at a dose of 400 mg/kg were nearly much like that of standard drug paracetamol. The results were recorded as mean ± SD. Out of all the groups apart from negative control, the temperature became normal through 3 h of study (Figure 1, Table2).
Subcutaneous injection of the pyrogenic dose of yeast produced elevated changes in rectal temperature, which is shown in Table 2. The MELR and MEAC showed a significant (P<0.05) decrease in rectal temperature at dose 400 mg/kg when compared with the standard. Extract at doses of 200, and 400 mg/kg showed a progressive decline in mean temperature pattern with the increase in the dose. Paracetamol showed significant (P<0.001) decrease in rectal temperature. The onset of action of paracetamol was 30 min. The line graph (Fig. 1) shows that paracetamol registered a phenomenal decrease in mean temperature from 38.7±0.12 to 37.01±0.42. Figure 2 illustrates that the most significant (P < 0.05) decrease in round-the-clock mean temperature in this study was shown by paracetamol followed by MELR and MEAC at 400 mg/kg and 200 mg/kg. Statistical analysis was performed using one-way analysis of variance followed by Tukey's test. The consequences of extract at a dose of 400 mg/kg were nearly much like that of standard drug paracetamol. The results were recorded as mean ± SD. Out of all the groups apart from negative control, the temperature became normal through 3 h of study (Figure 1, Table2).
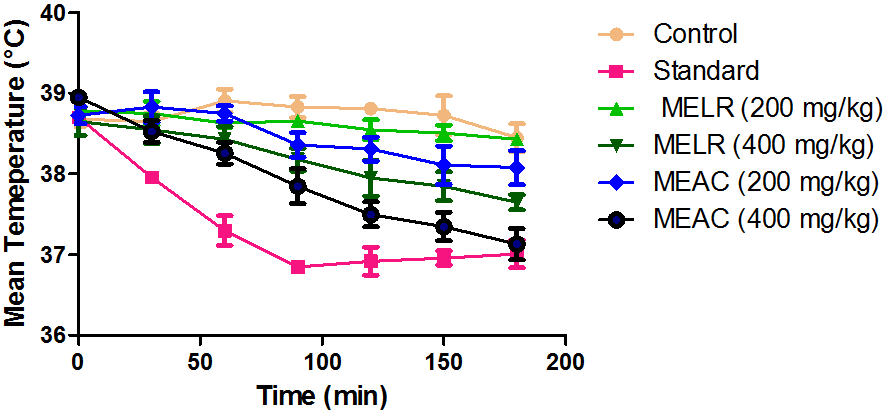
Figure 1: Line graph showing the effect of paracetamol and MELR and MEAC at 200 and 400 mg/kg on yeast-induced pyrexia in rats.
| Groups | BBT | Rectal Temperature (°C) after 10h of yeast injection | ||||||
| 0 min | 30 min | 60 min | 90 min | 120 min | 150 min | 180 min | ||
| Control | 37.12 ± 0.23 | 38.68 ± 0.21 | 38.65 ± 0.25 | 38.91 ± 0.35 | 38.83 ± 0.32 | 38.81 ± 0.18 | 38.73 ± 0.58 | 38.45 ± 0.43 |
| Standard (Paracetamol 150 mg/kg) | 37.16 ± 0.21 | 38.7 ± 0.12# | 37.96 ± 0.18# | 37.3 ± 0.45# | 36.85 ± 0.15# | 36.92 ± 0.43# | 36.96 ± 0.22# | 37.01 ± 0.42# |
| MELR (200 mg/kg) | 37.25 ± 0.19 | 38.78 ± 0.33a | 38.75 ± 0.37a | 38.63 ± 0.13a | 38.66 ± 0.12a | 38.55 ± 0.3a | 38.51 ± 0.23a | 38.43 ± 0.16a |
| MELR (400 mg/kg) | 37.18 ± 0.22 | 38.65 ± 0.42b | 38.55 ± 0.23b | 38.43 ± 0.39b | 38.18 ± 0.35b | 37.95 ± 0.54b | 37.85 ± 0.44b | 37.65 ± 0.23b |
| MEAC (200 mg/kg) | 37.08 ± 0.16 | 38.73 ± 0.26 | 38.83 ± 0.46 | 38.75 ± 0.23 | 38.36 ± 0.37 | 38.31 ± 0.36 | 38.11 ± 0.58 | 38. 08 ± 0.52 |
| MEAC (400 mg/kg) | 37.16 ± 0.13 | 38.95 ± 0.18b | 38.53 ± 0.33b | 38.26 ± 0.33b | 37.85 ± 0.53b | 37.5 ± 0.38b | 37.35 ± 0.43b | 37.13 ± 0.48b |
Table 2: Effect of MELR and MEAC on yeast extract induced pyrexia in Rats. All values expressed as mean±SEM; n=6 rats in each group, by one-way ANOVA followed by Tukey’s Multiple Comparison Test. #, p < 0.001 Vs Paracetamol (150 mg/Kg); a, p < 0.01 Vs Paracetamol (150 mg/Kg) and b, p < 0.05 Vs Control.
Discussion
Fever is the main defensive reaction referred to as the "acute phase reaction", which happen throughout the inflamed processes of various sources. Brewer's yeast (lipopolysaccharide that is the cell wall element of Gram-negative bacteria) is an exogenous pyrogen which binds to the immunological protein referred to as lipopolysaccharide binding protein. This kind of binding leads to the synthesis as well as the release of numerous endogenous cytokine factors, for example, interleukin (IL)-1, IL-6, and TNFa, that trigger the arachidonic acid pathway, and also eventually result in the synthesis and release of prostaglandin E2 (PGE2). Yeast-induced pyrexia is termed as pathogenic fever [17].
Based on the traditional perspective, fever is evoked through inflamed mediators (I L-1, IL-2, TNFα, others) unveiled through the peripheral mononuclear macrophages and other immune cells [18]. These fever-promoting cytokines tend to be moved through the bloodstream to the brain by particular carriers [19]. Cytokines are carried through the blood stream and also enter the brain through the circumventricular organs [20]. On the other hand, the cytokines might interact with their receptors on brain endothelial cells or perivascular tissue [21]. This presumed mechanism of fever induction is called the humoral hypothesis of fever induction. These pro-inflammatory mediators address the preoptic/anterior hypothalamus activating the release of PGE2 made out of cyclooxygenase (COX-2), and therefore increasing the body temperature [22].
In this study, orally administered paracetamol at 150 mg/kg significantly attenuated baker's yeast-induced fever in rats. Our study results are matching to other studies that have also shown the reduction of temperature in rats by paracetamol at the same dose. Antipyretics and non-steroidal anti-inflammatory drugs (NSAIDS) reduce temperature by inflammation reduction in the peripheral and CNS thermoregulatory sites.
In the current study, L. repens and A. cymosa methanolic extracts reduced baker's yeast-induced fever in rats significantly. The preliminary phytochemical study of both plants revealed the presence of flavonoids, alkaloids, tannins, phenols, steroids, glycosides, amino acids, and proteins majorly. The presence of these bioactive compounds may be responsible for the antipyretic activity of these extracts as sterols like β-Sitosterol [23] have the antipyretic effect. Alkaloids such as bolidine can reduce the raised temperature by suppressing the prostaglandin E2 synthesis [24]. In the same manner, flavonoids such as baicalin have the antipyretic outcome by curbing TNF- α [25].
It could be assumed that MELR andMEAC have antipyretic effect by reducing the concentration of PGE2 in the hypothalamus or by interrupting the steps that connect the peripheral inflammation with the central production of PGE2 or both [26].
Conclusion
It is concluded that MELR and MEAC have significant antipyretic activity. The traditional use of L. repens and A. cymosa in fever is supported by this study, and it would encourage its use in fever with the greater degree of assurance of its efficacy. It is recommended to determine the active chemical constituents accountable for the antipyretic activity.
References
- Metrouh-Amir H., et al. "Evaluation in vivo of anti-inflammatory and analgesic properties of Matricaria pubescens alkaloids". South African Journal of Botany 116 (2018): 168-174.
- Husseini Y., et al. "Analgesic and anti-inflammatory activities of hydro-alcoholic extract of Lavandula officinalis in mice: possible involvement of the cyclooxygenase type 1 and 2 enzymes". Revista Brasileira de Farmacognosia 26.1 (2016): 102-108.
- Les DH. "Aquatic dicotyledons of North America: ecology, life history, and systematics: CRC Press". (2017).
- Hsu H., et al. "Oriental materia medica: a concise guide: Keats Publishing (1996).
- Pullaiah T. "Encyclopaedia of world medicinal plants: Daya books" (2006).
- Brahmachari G. "Limnophila (Scrophulariaceae): Chemical and Pharmaceutical Aspects-An Update". The Open Natural Products Journal 7.4 (2014).
- Do QD., et al. "Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica". Journal of food and drug analysis 22 (204): 296-302.
- Kukongviriyapan U., et al. "Antioxidant and vascular protective activities of Cratoxylum formosum, Syzygium gratum and Limnophila aromatica". Biological and Pharmaceutical Bulletin 30(2007): 661-666.
- Rao JV., et al. "Antimicrobial activity of the essential oil of Limnophila gratissima". Fitoterapia 60.4 (1989): 376-377.
- Nanasombat S., et al. "Antimicrobial, antioxidant and anticancer activities of Thai local vegetables". Journal of Medicinal Plants Research 3.5(2009): 443-449.
- Suksamrarn A., et al. "Antimycobacterial and antioxidant flavones from Limnophila geoffrayi". Archives of pharmacal research 26.10 (2003): 816-820.
- Marles RJ., et al. "Antidiabetic plants and their active constituents". Phytomedicine 2.2 (1995): 137-189.
- Galani VJ., et al. "Psychotropic activity of Argyreia speciosa roots in experimental animals". Ayu 32.2 (2011): 380.
- Packialakshmi N., et al. "Antibacterial Screening on Leaves of Argyreia Cymosa Roxb. Against Pathogenic Bacteria Isolated from Infected Pateints Samples Wound, Sputum and Stool". International Journal of Applied Sciences and Biotechnology 2(3): 279-282.
- Kiran PM., et al. "Investigation of hepatoprotective activity of Cyathea gigantea (Wall. ex. Hook.) leaves against paracetamol-induced hepatotoxicity in rats". Asian Pacific journal of tropical biomedicine 2.5 (2012): 352.
- Vogel HG. "Drug discovery and evaluation: pharmacological assays: Springer Science & Business Media". (2002).
- Bhattacharya A., et al. "Antipyretic effect of ethanolic extract of Moringa oleifera leaves on albino rats". Tanta Medical Journal 42.2 (2014): 74.
- Zeisberger E. From humoral fever to neuroimmunological control of fever". Journal of Thermal Biology 24.5 (1999): 287-326.
- Banks WA., et al. "Permeability of the blood-brain barrier to soluble cytokine receptors". Neuroimmunomodulation 2.3(1995):161-165.
- Roth J., et al. "Signaling the brain in systemic inflammation: role of sensory circumventricular organs". Frontiers in Bioscience 9.7(2004): 290-300.
- Schiltz JC., et al. "Signaling the brain in systemic inflammation: the role of perivascular cells". Frontiers in Bioscience 8 (2003): 1321-1329.
- Saper CB., et al. "The neurologic basis of fever". The New England Journal of Medicine 330.26 (1994): 1880-1886.
- Gupta M., et al. "Anti-inflammatory and antipyretic activities of β-sitosterol". Planta medica 39.6 (1980): 157-163.
- Backhouse N., et al. "Anti-inflammatory and antipyretic effects of boldine". Agents and actions 42.3 (1994): 114-117.
- Chang C-P., et al. "flavonoid baicalin protects against cerebrovascular dysfunction and brain inflammation in experimental heatstroke". Neuropharmacology 52.2(2007): 1024-1033.
- Li S., et al. "Acetaminophen: antipyretic or hypothermic in mice? In either case, PGHS-1b (COX-3) is irrelevant". Prostaglandins Other Lipid Mediat. 85.5 (2008): 89-99.
Citation:
Venkateswarlu Gunji and Ganapathy S. “Antipyretic Effect of Methanolic Extracts of Limnophila repens and Argyreia cymosa
Whole Plant on Albino Rats”. Chronicles of Pharmaceutical Science 3.1 (2018): 753-759.
Copyright: © 2018 Venkateswarlu Gunji and Ganapathy S. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.