Research Article
Volume 3 Issue 1 - 2018
Enlighten the Treatment Strategies with Dietary Fibers of Rare Hirschsprung’s Disease Over Surgical Procedure
Department of Pharmaceutical Technology, Dr. APJ Abdul Kalam Technical University, India
*Corresponding Author: Firoz Khan, M. Pharm. (Pharmacology), Department of Pharmaceutical Technology (MIET), NH-58, Meerut, Uttar Pradesh, India.
Received: August 30, 2018; Published: September 15, 2018
Abstract
Hirschsprung disease is a condition in which mostly the neonates are more affected than adults but later it can be happens in old age of the patients. It can be developed due to malnutrition during pregnancy, genetic predisposing or change in the lifestyle, less physical exercise, reduce intake of fluids and fibers leading to destruction of ganglionic cells in the intestine. The first stage of Hirschsprung disease starts with constipation and irritable bowel syndrome leading to inflammation in the stomach with reduced and hard passage of stool from the anus. If the laxative fails to effect continuously, surgery would be preferred for last option. But after surgery there are several side effects are reported which creates the patient compliance such as diarrhea, vomiting, stomach pain, bleeding from anus and sometimes reduce immunity. The use of natural occurring medication helps to treat and manage the rare Hirschsprung disease (HSCR). Fibers are natural occurring component for improving the condition of HSCR disease. These fibers can be prepared by whole grains, psyllium seeds, flaxseeds, green vegetables, fruits and nuts. The more amount of taking fibers increases the absorption of water in intestine because it contains more water to absorb. Fibers also increase colon transit by decreasing the amount of luminal pH. It is more reliable with better patient compliance treatment with very less side effect compare to surgery. Patient has advised to take proper amount of fibers in the diet to reduce the symptoms of the disease. The diet should be divided in 4 or 5 small meals in a day to produce it effect.
Keywords: Hirschsprung disease; Plant fibers; Psyllium Husk and Laxatives
Abbreviations: Hirschsprung disease (HSCR)
Introduction
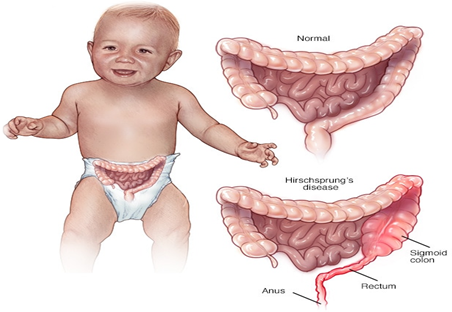
Hirschsprung disease is a relatively rare condition, initially described as a cause of constipation in early infancy as well as in some adults, and it is managed by pediatric surgeons [Bakari., et al. 2012]. Hirschsprung’s disease is the commonest cause of functional intestinal obstruction in the newborn and in some series is the second commonest cause of intestinal obstruction in neonates following anorectal malformations [Ameh and Chiradab., et al. 2000]. The management of this condition to create a colostomy to relief the intestinal obstruction while allowing for nutritional rehabilitation of the patient and then a formal pull through before the closure of the stoma [Katayoon., et al. 2005]. With the treatment of Hirschsprung disease the patient suffered from prolonged hospital stay; morbidity and mortality. [Santos., et al. 1999].
In the last few decades, attempts have been made by several workers to achieve a pull through without creating a stoma [Langer., et al. 2003]. In other sense, Hirschsprung disease (HSCR) or congenital intestinal aganglionosis is characterized by complete absence of neuronal ganglion cells from a portion of the intestinal tract, most commonly in the large intestine. The main sign or symptom of HSCR is constipation usually appearing shortly after birth. This constipation is chronic in nature and usually not relieved with laxatives. The present case is of a patient having HSCR which was successfully managed with Ayurvedic treatment [Kenny., et al. 2010].
Sign and symptoms of Hirschsprung disease
Swollen belly
Vomiting, including vomiting a green or brown substance
Constipation or gas, which might make a newborn fussy
Diarrhea
Swollen belly
Vomiting, including vomiting a green or brown substance
Constipation or gas, which might make a newborn fussy
Diarrhea
Causes
The most accepted theory of the cause of Hirschsprung disease (HSCR) is that there is a defect in the craniocaudal migration of neuroblasts originating from the neural crest, a process that begins at four weeks of gestation and ends at week 7 with the arrival of neural crest-derived cells at the distal end of the colon [Fu M., et al. 2004]. Failure of the cells to reach the distal colon leaves that segment aganglionic and therefore nonfunctional, resulting in Hirschsprung disease. Defects in the differentiation of neuroblasts into ganglion cells and ganglion cell destruction within the intestine may also contribute to the disorder [McKeown., et al. 2013].
The most accepted theory of the cause of Hirschsprung disease (HSCR) is that there is a defect in the craniocaudal migration of neuroblasts originating from the neural crest, a process that begins at four weeks of gestation and ends at week 7 with the arrival of neural crest-derived cells at the distal end of the colon [Fu M., et al. 2004]. Failure of the cells to reach the distal colon leaves that segment aganglionic and therefore nonfunctional, resulting in Hirschsprung disease. Defects in the differentiation of neuroblasts into ganglion cells and ganglion cell destruction within the intestine may also contribute to the disorder [McKeown., et al. 2013].
Complications
Children who have Hirschsprung's disease are prone to a serious intestinal infection called enterocolitis. Enterocolitis can be life-threatening. It's treated in the hospital with colon cleaning and antibiotics [Goldstein.,
Children who have Hirschsprung's disease are prone to a serious intestinal infection called enterocolitis. Enterocolitis can be life-threatening. It's treated in the hospital with colon cleaning and antibiotics [Goldstein.,
Diagnosis
Abdominal X-ray using a contrast dye
Barium or another contrast dye is placed into the bowel through a special tube inserted in the rectum. The barium fills and coats the lining of the bowel, creating a clear silhouette of the colon and rectum. The X-ray will often show a clear contrast between the narrow section of bowel without nerves and the normal but often swollen section of bowel behind it [Amiel J and Sproat-Emison., et al. 2008].
Abdominal X-ray using a contrast dye
Barium or another contrast dye is placed into the bowel through a special tube inserted in the rectum. The barium fills and coats the lining of the bowel, creating a clear silhouette of the colon and rectum. The X-ray will often show a clear contrast between the narrow section of bowel without nerves and the normal but often swollen section of bowel behind it [Amiel J and Sproat-Emison., et al. 2008].
Measuring control of the muscles around the rectum
A manometry test is typically done on older children and adults. The doctor inflates a balloon inside the rectum. The surrounding muscle should relax as a result due to Hirschsprung's disease [Moore., et al. 2009].
A manometry test is typically done on older children and adults. The doctor inflates a balloon inside the rectum. The surrounding muscle should relax as a result due to Hirschsprung's disease [Moore., et al. 2009].
Biopsy
This is the surest way to identify Hirschsprung's disease. A biopsy sample can be collected using a suction device, then examined under a microscope to determine whether nerve cells are missing. After confirmation of nerve cell damage it will go for treatment (Fitze., et al. 2002).
This is the surest way to identify Hirschsprung's disease. A biopsy sample can be collected using a suction device, then examined under a microscope to determine whether nerve cells are missing. After confirmation of nerve cell damage it will go for treatment (Fitze., et al. 2002).
Treatment by surgery
Surgery to bypass the part of the colon that has no nerve cells treats Hirschsprung's disease. The lining of the diseased part of the colon is stripped away, and normal colon is pulled through the colon from the inside and attached to the anus. This is usually done using minimally invasive (laparoscopic) methods, operating through the anus [Kim., et al. 2006].
Surgery to bypass the part of the colon that has no nerve cells treats Hirschsprung's disease. The lining of the diseased part of the colon is stripped away, and normal colon is pulled through the colon from the inside and attached to the anus. This is usually done using minimally invasive (laparoscopic) methods, operating through the anus [Kim., et al. 2006].
In children who are very ill, surgery might be done in two steps. First, the abnormal portion of the colon is removed and the top, healthy portion of the colon is connected to an opening the surgeon creates in the child's abdomen. Stool then leaves the body through the opening into a bag that attaches to the end of the intestine that protrudes through the hole in the abdomen (stoma). This allows time for the lower part of the colon to heal [Emison., et al. 2005]. Ostomy procedures include:
- Ileostomy: The doctor removes the entire colon and connects the small intestine to the stoma. Stool leaves the body through the stoma into a bag.
- Colostomy: The doctor leaves part of the colon intact and connects it to the stoma. Stool leaves the body through the end of the large intestine. Later, the doctor closes the stoma and connects the healthy portion of the intestine to the rectum or anus [Burzynski., et al. 2004].
Side effects of surgery
After surgery, most children pass stool normally — although some may have diarrhea at first. Toilet training may take longer because children have to learn how to coordinate the muscles seed to pass stool. Long term, it's possible to have continued constipation, a swollen belly and leaking of stool (soiling) (Uesaka., et al. 2007).
After surgery, most children pass stool normally — although some may have diarrhea at first. Toilet training may take longer because children have to learn how to coordinate the muscles seed to pass stool. Long term, it's possible to have continued constipation, a swollen belly and leaking of stool (soiling) (Uesaka., et al. 2007).
Children continue to be at risk of developing a bowel infection (enterocolitis) after surgery.
- Diarrhea
- Fever
- Swollen abdomen
- Vomiting
- Bleeding from the rectum. (Uesaka and Nagashimada., 2008)
Treatment with dietary fibers
Dietary fiber refers to the edible parts of plants or carbohydrates that cannot be digested. Fiber is in all plant foods, including fruits, vegetables, grains, nuts, seeds, and legumes. If the child has constipation after surgery, following fibers are can be preferred for relief-
Dietary fiber refers to the edible parts of plants or carbohydrates that cannot be digested. Fiber is in all plant foods, including fruits, vegetables, grains, nuts, seeds, and legumes. If the child has constipation after surgery, following fibers are can be preferred for relief-
- Add high-fiber food in diet: If the child eats solid foods, include high-fiber foods. Offer whole grains, fruits and vegetables and limit white bread and other low-fiber foods. Because a sudden increase in high-fiber foods can worsen constipation at first, add high-fiber foods to your child's diet slowly [annot., et al. 2013].
- Increase fluids: Encourage the child to drink more water. If a portion or the child’s entire colon was removed, your child may have trouble absorbing enough water. Drinking more water can help the child stay hydrated, which helps to easy pass the stool [Martucciello., et al. 2000].
- Encourage physical activity: Daily aerobic activity helps promote regular bowel movements.
- Laxatives: If the child does not respond to or cannot tolerate increased fiber, water or physical activity, certain laxatives [ex- Lectulose, Besacodyl etc.] medications to encourage bowel movements — might help relieve constipation [Bajaj., et al. 2005].
Besides that, several herbal drugs are also available for the treatment and management of Hirschsprung disease. These drug include-
Psyllium
Psyllium is a type of soluble fiber that comes from a shrub like herb called Planto ovata. Psyllium may be the most used fiber supplement on the market. Psyllium can be used in the conditions including hemorrhoids, irritable bowel syndrome, constipation and diarrhea. Psyllium husks can be consumed in their whole form either by mixing in fruit juice or milk. One glass of liquid can be mixed with dried psyllium seed to produce it effect. However, psyllium supplements are commonly ground into a more easily ingested powdered form [Garcia-Barcelo., et al. 2009].
Psyllium is a type of soluble fiber that comes from a shrub like herb called Planto ovata. Psyllium may be the most used fiber supplement on the market. Psyllium can be used in the conditions including hemorrhoids, irritable bowel syndrome, constipation and diarrhea. Psyllium husks can be consumed in their whole form either by mixing in fruit juice or milk. One glass of liquid can be mixed with dried psyllium seed to produce it effect. However, psyllium supplements are commonly ground into a more easily ingested powdered form [Garcia-Barcelo., et al. 2009].
Glucomannan
Glucomannan is a precursor of sugar that present in several plants including dietary fiber. It is derived from the root of the konjac plant. Glucomannan swells to about 17 times its original volume when immersed in water and used in the treatment of constipation. For this reason, glucomannan can be an effective bulking fiber in small doses [Pini Prato., et al. 2013].
Glucomannan is a precursor of sugar that present in several plants including dietary fiber. It is derived from the root of the konjac plant. Glucomannan swells to about 17 times its original volume when immersed in water and used in the treatment of constipation. For this reason, glucomannan can be an effective bulking fiber in small doses [Pini Prato., et al. 2013].
Methylcellulose
Methylcellulose is a bulk-forming fiber that can increase the amount of water in stool, which can help relieve constipation. As with any bulk-forming fiber supplement, it is important to take methylcellulose with a full glass of water. Methylcellulose is the key ingredient in a popular brand of fiber supplement [Menezes., et al. 2005].
Methylcellulose is a bulk-forming fiber that can increase the amount of water in stool, which can help relieve constipation. As with any bulk-forming fiber supplement, it is important to take methylcellulose with a full glass of water. Methylcellulose is the key ingredient in a popular brand of fiber supplement [Menezes., et al. 2005].
Flaxseed
In addition to being high in fiber, flaxseed also possesses other beneficial properties including rich concentrations of omega-3 fatty acids, phytochemicals and lignans. Flaxseeds can be purchased in their whole form or in a ground supplement, which may be easier for the body to digest. The use of constipation in these seeds is also used to treat Hirschsprung disease by several doctors [Bull., et al. 2011].
In addition to being high in fiber, flaxseed also possesses other beneficial properties including rich concentrations of omega-3 fatty acids, phytochemicals and lignans. Flaxseeds can be purchased in their whole form or in a ground supplement, which may be easier for the body to digest. The use of constipation in these seeds is also used to treat Hirschsprung disease by several doctors [Bull., et al. 2011].
Others
- Wheat dextrin is a natural fiber used in one popular brand of fiber supplement.
- Calcium polycarbophil is a bulk-forming fiber laxative used in another popular brand of fiber supplement [Raveenthiran., et al. 2011].
- Inulin is a starch like substance found in a variety of fruits, vegetables and herbs. Inulin supports the growth of a particular type of bacteria that may help improve bowel function [Parisi., et al. 2000].
Hirschsprung disease can be managed by adding the calculated fibers fruits in the diet. These foods are listed below in the table-
| Fruits | Serving size | Total fiber (grams)* |
| Raspberries | 1 cup | 8.0 |
| Pear, with skin | 1 medium | 5.5 |
| Apple, with skin | 1 medium | 4.4 |
| Banana | 1 medium | 3.1 |
| Orange | 1 medium | 3.1 |
| Strawberries (halves) | 1 cup | 3.0 |
| Figs, dried | 2 medium | 1.6 |
| Raisins | 1 ounce (60 raisins) | 1.0 |
Table 1: List of fruits useful in the Hirschsprung disease.
In other hand the whole grains, certain cereal and pastas are also useful in relieving symptoms of Hirschsprung disease. These foods provide proper nutrition and extent of water in the intestine to absorb more water to obtain desired osmotic pressure to easy release the stool. These foods can be eat in breakfast, mid lunch and evening.
| Grains, cereal and pasta | Serving size | Total fiber (grams) |
| Spaghetti, whole-wheat, cooked | 1 cup | 6.3 |
| Barley, pearled, cooked | 1 cup | 6.0 |
| Bran flakes | 3/4 cup | 5.5 |
| Oat bran muffin | 1 medium | 5.2 |
| Oatmeal, instant, cooked | 1 cup | 4.0 |
| Popcorn, air-popped | 3 cups | 3.6 |
| Brown rice, cooked | 1 cup | 3.5 |
| Bread, rye | 1 slice | 1.9 |
| Bread, whole-wheat | 1 slice | 1.9 |
Table 2: Management of Hirschsprung disease by using listed breakfast.
Nuts, seeds and certain legumes provide beneficial protein and fibers to the health of the patient as well as to treat the early stages of Hirschsprung disease.
| Legumes, nuts and seeds | Serving size | Total fiber (grams)* |
| Split peas, boiled | 1 cup | 16.3 |
| Lentils, boiled | 1 cup | 15.6 |
| Black beans, boiled | 1 cup | 15.0 |
| Lima beans, boiled | 1 cup | 13.2 |
| Baked beans, vegetarian, canned, cooked | 1 cup | 10.4 |
| Almonds | 1 ounce (23 nuts) | 3.5 |
| Pistachio nuts | 1 ounce (49 nuts) | 2.9 |
| Pecans | 1 ounce (19 halves) | 2.7 |
Table 3: List of available nuts and seeds for treating the HSCR disease.
Although, vegetables plays a major role in the management of several disease but in case of HSCR, the fibers of mostly green veggies relief the symptoms of constipation and irritable bowel syndrome. Green vegetables retain water in the intestine and its fibers reduce the hardness of stool. With this property, doctors advice to eat more green leafy vegetables to lower the symptoms of HSCR [Feldmen., et al. 2002].
| Vegetables | Serving size | Total fiber (grams)* |
| Artichoke, boiled | 1 medium | 10.3 |
| Green peas, boiled | 1 cup | 8.8 |
| Broccoli, boiled | 1 cup | 5.1 |
| Turnip greens, boiled | 1 cup | 5.0 |
| Brussels sprouts, boiled | 1 cup | 4.1 |
| Sweet corn, boiled | 1 cup | 3.6 |
| Potato, with skin, baked | 1 small | 2.9 |
| Tomato paste, canned | 1/4 cup | 2.7 |
| Carrot, raw | 1 medium | 1.7 |
Table 4: Vegetables with high amount of fibers.
Mechanism of action of fibers
The mechanism of action of fiber on constipation includes:
The mechanism of action of fiber on constipation includes:
- Fiber increases stool bulk and accelerates colon transit.
- Fermenting fiber produces short-chain fatty acids (butyrate, propionate, acetate, etc.), which increase osmotic load and accelerate colon transit [Stewart., et al. 2003].
- Short-chain fatty acids change the intraluminal microbiome (mass) directly or indirectly by decreasing luminal pH, which accelerates colon transit [Holschneider., et al. 2003].
- Fiber contains more water compare to other food [Coran., et al. 2000].
All these mechanism improve stool consistency and amount. Classically, fiber is classified into water-soluble and water-insoluble fiber. Water-insoluble fiber includes cellulose, hemicellulose, methylcelluose, lignin, and synthetic fibers (calcium polycarbophil). Water-soluble fiber includes gums (fenugreek gum, guar gum, tara gum, locust bean gum, or carob gum), pectin, mucilage, psyllium, and glucomannan [Pakarinen., et al. 2005].
Results and Discussion
Hirschsprung disease is a rare, chronic and preventable condition that affected 5% over 1000 children’s. Later it can be happens in adult age with lack of nutrition and unproven lifestyle adopted leading to long time constipation and hyperacidity. Some of the treatments are available to treat HSCR disease but they should not recover the patient well; needs surgery at the end. The use of calculated diet with rich of fluids and fibers including psyllium husk, methylcellulose, glucomannan, green leafy veggies, nuts, whole grains and some fruits reduce the risk of surgery (colostomy) and relief for longer time but if the desired amount of food will not be ingested it will needs surgery at the last option of the treatment.
Conclusion
By the use of fibers fruits, vegetables, nuts, and proper dose of natural laxatives (Psyllium, Senna leaf, castor oil, and methylcellulose) lower the risk of Hirschsprung disease in the future. It is easy, inexpensive and less time consuming process to treat this rare disease of Hirschsprung disease. These can be made easy in the every kitchen and useful in growing countries like India, China and Africa with more cases compare to developed countries.
Acknowledgements
I would like to thanks Dr. APJ Abdul kalam Techniccal University, Uttar Pradesh, Lucknow to do this review article with encouragement and push up my adrenaline level to believe myself for submitting this article.
I would like to thanks Dr. APJ Abdul kalam Techniccal University, Uttar Pradesh, Lucknow to do this review article with encouragement and push up my adrenaline level to believe myself for submitting this article.
Conflict of interest
The author declares no conflict of interest
The author declares no conflict of interest
References
- Ameh EA., et al. “Neonatal intestinal obstruction in Zaria, Nigeria”.East African Medical Journal 9 (2000): 510-513.
- Amiel J., et al. “Hirschsprung disease, associated syndromes and genetics: a review”. Journal Of Medical Genetics45 (2008): 47-52.
- Bull MJ., et al. “Committee on Genetics. Health supervision for children with Down syndrome”. Pediatrics 128 (2011): 392-393.
- Bakari AA., et al. “Congenital aganglionic megacolon in Nigerian adults: two case reports and review of the literature”. Nigerian journal of clinical practice 14 (2011): 249-252.
- Burzynski GM., et al. “Localizing a putative mutation as the major contributor to the development of sporadic Hirschsprung disease to the RET genomic sequence between the promoter region and exon 2”. European Journal of Human Genetics 12 (2004): 604-607.
- Bajaj R., et al. “Congenital central hypoventilation syndrome and Hirschsprung's disease in an extremely preterm infant”. Pediatrics 115 (2005): 737-740.
- Coran AG., et al. “Recent advances in the management of Hirschsprung’s disease”. The American Journal of Surgery180 (2000): 382-387.
- Emison ES., et al. “A common sex-dependent mutation in a RET enhancer underlies Hirschsprung disease risk”.Nature 434 (2005): 855-857.
- Fitze G., et al. “Association between c135G/A genotype and RET proto-oncogene germline mutations and phenotype of Hirschsprung's disease”. Lancet 359 (2002): 1200.
- Fu M., et al. “Embryonic development of the ganglion plexuses and the concentric layer structure of human gut: a topographical study”. Anatomy and Embryology208 (2004): 28-33.
- Feldmen M., et al. “Hirschsprung’s disease: congenital megacolon. In: Sleisenger & Fordtran’s Gastrointestinal and Liver Disease: Pathophysiology, Diagnosis, Management. Philadelphia” Pa Saunders (2002): 2131-2135.
- Garcia-Barcelo MM., et al. “Genome-wide association study identifies NRG1 as a susceptibility locus for Hirschsprung's disease”. Proceedings of the National Academy of Sciences106 (2009): 2690-2694.
- Goldstein AM., et al. “Building a brain in the gut: development of the enteric nervous system”. Clinical Genetics83 (2013): 301-307.
- Holschneider AM., et al. “Hirschsprung’s Disease and Allied Disorders”. Harwood Academic Publishers (2000): 134-138.
- Jannot AS., et al. “Chromosome 21 scan in Down syndrome reveals DSCAM as a predisposing locus in Hirschsprung disease”. PLoS One 8 (2013): 625-631.
- Kim JH., et al. “Novel mutations of RET gene in Korean patients with sporadic Hirschsprung's disease”. Journal of Pediatric Surgery 41 (2006): 1245-1250.
- Katayoon Shayan., et al. “Reliability of intraoperative frozen sections in the management of Hirschsprung’s disease”. Journal of Pharmacology and Pharmacotherapeutics 18 (2014): 85-92.
- Kenny SE., et al. “Hirschsprung's disease”. Seminars in Pediatric Surgery19 (2010): 194-200.
- Langer JC., et al. “One-stage transanal Soave pull-through for Hirschsprung disease: A multicenter experience with 141 children”. Annals of Surgery 2003; 238:569-583. Discussion 583–585.
- McKeown SJ., et al. “Hirschsprung disease: a developmental disorder of the enteric nervous system”. Wiley Interdisciplinary Reviews-Developmental Biology Journal2 (2013): 108-113.
- Moore SW., et al. “Clinical and genetic differences in total colonic aganglionosis in Hirschsprung's disease”. Journal of Pediatric Surgery44 (2009): 1899-1905.
- Martucciello G., et al. “Pathogenesis of Hirschsprung's disease”. Journal of Pediatric Surgery35 (2000): 1010-1017.
- Menezes M., et al. “Long-term clinical outcome in patients with Hirschsprung's disease and associated Down's syndrome”. Journal of Pediatric Surgery 40 (2005): 810-818.
- Pakarinen MP., et al. “Increased incidence of medullary thyroid carcinoma in patients treated for Hirschsprung’s disease”. Journal of Pediatric Surgery 40 (2005): 1532-1534.
- Parisi MA., et al. “Genetics of Hirschsprung disease”. Current Opinion in Pediatrics 12 (2000): 610-617.
- Pini Prato A., et al. “A prospective observational study of associated anomalies in Hirschsprung's disease”. Orphanet Journal of Rare Diseases 8 (2013): 184-191.
- Raveenthiran V. “Knowledge of ancient Hindu surgeons on Hirschsprung disease: evidence from Sushruta Samhita of circa 1200-600 bc”. Journal of Pediatric Surgery46 (2011): 2204-2208.
- Stewart DR., et al. “The genetics of Hirschsprung disease”. Gastroenterology Clinics of North America 32 (2003): 819-837.
- Santos MC., et al. “Primary Swenson pull-through compared with multiple-stage pull-through in the neonate”. Journal of Pediatric Surgery34 (1999): 1079-1081.
- Uesaka T., et al. “Conditional ablation of GFRalpha1 in postmigratory enteric neurons triggers unconventional neuronal death in the colon and causes a Hirschsprung's disease phenotype”. Development 134 (2007): 2171-2176.
- Uesaka T., et al. “Diminished Ret expression compromises neuronal survival in the colon and causes intestinal aganglionosis in mice”. Journal of Clinical Investigation 118 (2008): 1890-1897.
Citation:
Firoz Khan. “Enlighten the Treatment Strategies with Dietary Fibers of Rare Hirschsprung’s Disease Over Surgical Procedure”.
Chronicles of Pharmaceutical Science 3.1 (2018): 737-744.
Copyright: © 2018 Firoz Khan. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.