Research Article
Volume 1 Issue 5 - 2017
Synthesis and Characterization of Bactericidal Agents Derived from Neem (Azadirecta Indica) Oil
1Department of Chemistry, SPC Govt. College Ajmer
2Department of Biochemistry, Dr. S.N. Medical College Jodhpur
2Department of Biochemistry, Dr. S.N. Medical College Jodhpur
*Corresponding Author: Priyanka Kumawat, Department of Chemistry, SPC Govt. College Ajmer.
Received: November 15, 2017; Published: December 06, 2017
Abstract
In this study, we evaluated the antimicrobial potency of copper (II) soap and copper soap complex synthesized/formulated in our laboratory. The solid copper (II) soap derived from Neem (AzadirectaIndica) oil and its complex with ligand containing nitrogen and oxygen atoms like urea has been synthesized and characterised by elemental analysis, IR spectroscopy and biological studies. From the analytical data, the stoichiometry of the complex has been observed to be 1:1 (metal: ligand). The bacterial studies of soap and complex have been investigated. The derived compound was found active against Staphylococcus aureus, Coagulase-negative staphylococci(CoNs), Acinetobacter baumanii,Pseudomonas aeruginosaand micrococcusbacteria. These findings have high medical, industrial and economic significance as copper (II) soap and copper (II) soap complex could be harnessed in the formulation of medicated soaps.
Keywords: Copper (II) soap; Complex; IR Spectroscopy; Biological studies
Introduction
According to Osbore and Grobe antibacterial soaps can remove 65 to 85% bacteria from human skin. Contemporary commercial antimicrobial soaps contain synthetic chemicals such as triclosan, tri-clorocarbanilide and chloroxylenol, most of which are thought to be car-cinogenic, mutagenic and or generate allergic reactions. Triclosan (2, 4, 4’trichloro-2’hydroxydiphenyl ester) has been used in soaps, shampoo and fabrics, as an antimicrobial agent [1].
Copper (II) soap derived from non-edible Neem (Azadirecta Indica) oil play a vital role in various fields due to their surface active properties. This oil was particularly chosen as it is easily available commercially and biodegradable in nature. Copper soap have a tendency of complexation with ‘nitrogen’ and ‘oxygen’ containing ligands. Using urea as a ligand, complexation of synthesized copper soap has been done to obtain its complex.
Since copper metal is toxic in nature, literature survey reveals that the synthesized copper soap and its urea complex may play a significant role in biological activities. The interest in co-ordination chemistry is increasing continuously with the preparation of organic ligands containing a variety of donor groups and it is multiplied many folds when the ligand have biological importance [2–4].
Copper metal as well as nitrogen and oxygen containing compounds play a vital role in fungicidal activities. Various non-edible oils are widely used, easily available, and are eco -friendly. Also, vegetable oil based lubricants are slowly replacing the mineral oils due to their extraordinary bio-degradability and many other specific properties [5-7]. These facts led us to synthesize copper soaps of non-edible oils and its complex with ligand containing nitrogen and oxygen elements, investigate the role of copper and other elements in biological activities. It is anticipated that it will generate new hopes in agrochemical, industrial and pharmacological field [8-11].
Micro-Organisms taken for study are as follow
Micro-organisms employed to study and explain the bactericidal processes of complex synthesized in our laboratory were as follows:
Micro-organisms employed to study and explain the bactericidal processes of complex synthesized in our laboratory were as follows:
Staphylococcus aureus (S. aureus)
Staphylococcus aureus is an anaerobic, Gram-positive cocci, which appears as grape-like clusters when viewed through a microscope and has large, round, golden-yellow colonies, often with hemolysis, when grown on blood agar plates. The golden appearance is the etymological root of the bacteria's name; aureus means "golden" in Latin. S. aureusmay occur as a commensal on human skin; it also occurs in the nose frequently (in about a third of the population) and throat less commonly [12-13].
Staphylococcus aureus is an anaerobic, Gram-positive cocci, which appears as grape-like clusters when viewed through a microscope and has large, round, golden-yellow colonies, often with hemolysis, when grown on blood agar plates. The golden appearance is the etymological root of the bacteria's name; aureus means "golden" in Latin. S. aureusmay occur as a commensal on human skin; it also occurs in the nose frequently (in about a third of the population) and throat less commonly [12-13].
Coagulase-negative staphylococci (CoNS)
The definition of the heterogeneous group of coagulase-negative staphylococci (CoNS) is still based on diagnostic procedures thatfulfill the clinical need to differentiate between Staphylococcus aureusand those staphylococci classified historically as being less or nonpathogenic. Due to patient- and procedure-related changes, CoNS now represent one of the major nosocomial pathogens, with S. epidermidisand S. haemolyticus being the most significant species.
The definition of the heterogeneous group of coagulase-negative staphylococci (CoNS) is still based on diagnostic procedures thatfulfill the clinical need to differentiate between Staphylococcus aureusand those staphylococci classified historically as being less or nonpathogenic. Due to patient- and procedure-related changes, CoNS now represent one of the major nosocomial pathogens, with S. epidermidisand S. haemolyticus being the most significant species.
They account substantially for foreign body-related infections and infections in preterm newborns. While S. saprophyticuhas been associated with acute urethritis, S. lugdunensishas a unique status, in some aspects resembling S. aureusin causing infectious endocarditis. In addition to CoNS found as food-associated saprophytes, many other CoNS species colonize the skin and mucous membranes of humans and animals and are less frequently involved in clinically manifested infections.
This blurred Coagulase-Negative Staphylococci gradation in terms of pathogenicity is reflected by species- and strain-specific virulence factors and the development of different host-defending strategies. Clearly, CoNS possess fewer virulence properties than S. aureus, with a respectively different disease spectrum. In this regard, host susceptibility is much more important. Therapeutically, CoNS are challenging due to the large proportion of methicillin-resistant strains and increasing numbers of isolates with less susceptibility to glycopeptides [14-15].
Acinetobacter baumanii
Acinetobacter species are saprophytic and ubiquitous and can be found in natural (e.g. soil, water, food) and hospital environment. Acinetobacter is considered as a part of commensal flora of man (e.g. axillae, groin, digit webs) where they occasionally present as opportunistic pathogens. Cutaneous colonization can be seen in approximately 25% of population. 7% of adults and children show transient pharyngeal colonization.
Acinetobacter species are saprophytic and ubiquitous and can be found in natural (e.g. soil, water, food) and hospital environment. Acinetobacter is considered as a part of commensal flora of man (e.g. axillae, groin, digit webs) where they occasionally present as opportunistic pathogens. Cutaneous colonization can be seen in approximately 25% of population. 7% of adults and children show transient pharyngeal colonization.
It is often difficult to distinguish between the colonization and the infection with this organism and hence attribute the exact morbidity and mortality associated with infections due to this organism. The other genomospecies are unnamed. DNA groups 1, 2, 3 and 13 are sachharolytic strains and are collectively referred to as Acinetobacter calcoaceticus-A. baumanniicomplex [16-17].
Pseudomonas aeruginosa (P. aeruginosa)
Pseudomonas aeruginosais an opportunistic pathogen, meaning that it exploits some break in the host defenses to initiate an infection. It is a Gram-negative, aerobic, rod-shaped bacterium with unipolar motility. The word Pseudomonas means false unit, from the Greek pseudo- (Greek: ψευδο, false) and Monas (Latin: Monas, from Greek: μονος, a single unit). The stem word Mon was used early in the history of microbiology to refer to germs, e.g., Kingdom Monera.
Pseudomonas aeruginosais an opportunistic pathogen, meaning that it exploits some break in the host defenses to initiate an infection. It is a Gram-negative, aerobic, rod-shaped bacterium with unipolar motility. The word Pseudomonas means false unit, from the Greek pseudo- (Greek: ψευδο, false) and Monas (Latin: Monas, from Greek: μονος, a single unit). The stem word Mon was used early in the history of microbiology to refer to germs, e.g., Kingdom Monera.
The species name aeruginosa is a Latin word meaning copper rust. This also describes the blue-green bacterial pigment seen in laboratory cultures of the species. This blue-green pigment is a combination of two metabolites of P. aeruginosa, pyocyanin (blue) and pyoverdine (yellow), which impart the blue-green characteristic color of cultures. Another assertion is that the word may be derived from the Greek prefix ae- meaning "old or aged, and the suffix ruginosa means wrinkledor bumpy [18-19].
Micrococcus
Micrococcus luteusis a gram positive, spherical, saprotrophic bacterium in obligate aerobe M. luteus is found in soil, dust, water and air and as part of the normal flora of the mammalian skin. The bacterium also colonies the human mouth, mucosae, oropharynx and upper respiratory tract. M. luteus is cogulase negative bacitracin susceptible and forms bright yellow colonies on nutrient agar. To confirm it is not Staphylococcus aureus a bacitracin susceptibility test performed [20-22].
Micrococcus luteusis a gram positive, spherical, saprotrophic bacterium in obligate aerobe M. luteus is found in soil, dust, water and air and as part of the normal flora of the mammalian skin. The bacterium also colonies the human mouth, mucosae, oropharynx and upper respiratory tract. M. luteus is cogulase negative bacitracin susceptible and forms bright yellow colonies on nutrient agar. To confirm it is not Staphylococcus aureus a bacitracin susceptibility test performed [20-22].
Experimental
All the chemicals used were of LR/AR grade. Solvent was purified according to standard procedures before use. Elemental analysis was done for soap and complex for their metal content following standard procedures. The complex under study was prepared in two steps. In the first step, copper soap was prepared and in the second step complexation of copper soap was done with ligand like urea [23]. Benzene was dehydrated by storage over sodium wire for 2–3 days and by refluxing for about twenty hours, it was then distilled and redistillation was carried out azeotropically with ethanol [24].
All the chemicals used were of LR/AR grade. Solvent was purified according to standard procedures before use. Elemental analysis was done for soap and complex for their metal content following standard procedures. The complex under study was prepared in two steps. In the first step, copper soap was prepared and in the second step complexation of copper soap was done with ligand like urea [23]. Benzene was dehydrated by storage over sodium wire for 2–3 days and by refluxing for about twenty hours, it was then distilled and redistillation was carried out azeotropically with ethanol [24].
Copper soap was prepared by refluxing the non-edible oil i.e. Neem (Azadirecta Indica) oil, in its pure form, of an available in the Indian market, with alcohol and 2N KOH solution for 3 hours (Direct metathesis). The neutralization of excess KOH present was done by 1N HCl. Saturated solution of copper sulphate was then added to it for conversion of neutralized soap into copper soap. Copper soap so obtained was then washed with hot water and dried. The soap was recrystallized using hot benzene. The fatty acid composition of the non-edible oil was confirmed through Gas liquid chromatography [GLC] of its methyl esters and is given in Table 1 [25-27].
| Name of oil | % Fatty acid | ||||
| 16:00 | 18:00 | 18:01 | 18:02 | Other acid (C20–C24) | |
| Neem oil | 14.9 | 14.4 | 61.9 | 7.5 | 1.3 |
Table 1: Fatty acid composition of oil used for copper soap/complex synthesis.
The ligand urea was taken directly. The purified copper soap derived from non-edible oil was refluxed with ligand (urea) in 1:1 ratio using benzene as a solvent for one hour. It was then filtered hot, dried, recrystallized and purified in hot benzene. Thin layer chromatography [TLC] using silica gel was used to check the purity of the complex [28-29].
The complex obtained was dark green in colour. The complex was soluble in benzene and other organic solvents but insoluble in water. The complex was stable at room temperature, its physical parameters like saponification value (S.V.), saponification equivalent (S.E.) and molecular weights are recorded in Table 2 On the basis of their elemental analysis, 1:1 (metal: ligand) type of stoichiometry has been suggested [30-32].
| Name of soap/complex | Colour | M.P. | Metal Content % | S.V. | S.E. | Average M.W. | |
| Observe | Calculate | ||||||
| Copper Neem Soap | Dark Green | 500c | 10.16 | 10.07 | 198 | 283.33 | 630.16 |
| Copper Neem Soap Urea Complex | Dark Green | 720c | 9.31 | 9.19 | - | - | 690.76 |
Table 2: Analytical and physical data of copper soap and its complex derived from Neem oil.
In order to study the structure of soap and complex, the infrared (IR) absorption spectra of compounds were obtained on a ABB Horizon MB 3000 series instrument spectrophotometer (4000–600 cm-1) from SPC Govt. College, Ajmer.
Antimicrobial Evaluation
The biological activities of copper soap and its corresponding complex with ligand urea have been screened against Staphylococcus aureus, Coagulase-negative staphylococci (CoNs), Acinetobacter baumanii, Pseudomonas aeruginosa and Micrococcusbacteria at 3 * 104 ppm, 1.5 * 104 ppm, .75 * 104 ppm and .375 * 104 ppm using disc of these solutions by Mueller Hinton Agar plates. The following Bacteria: Staphylococcus aureus, Pseudomonas aeruginosa, Cons, Micrococcus and Acinetobactor. All bacterial strains were maintained on nutrient agar medium at ±37°C. These cultures are obtained from the Department of Microbiology, Dr. S.N. Medical College, Jodhpur.
The biological activities of copper soap and its corresponding complex with ligand urea have been screened against Staphylococcus aureus, Coagulase-negative staphylococci (CoNs), Acinetobacter baumanii, Pseudomonas aeruginosa and Micrococcusbacteria at 3 * 104 ppm, 1.5 * 104 ppm, .75 * 104 ppm and .375 * 104 ppm using disc of these solutions by Mueller Hinton Agar plates. The following Bacteria: Staphylococcus aureus, Pseudomonas aeruginosa, Cons, Micrococcus and Acinetobactor. All bacterial strains were maintained on nutrient agar medium at ±37°C. These cultures are obtained from the Department of Microbiology, Dr. S.N. Medical College, Jodhpur.
The antimicrobial activity of newly synthesized compounds was evaluated using agar disc diffusion assay. Briefly, a 24 and 48 hours old culture of selected bacteria was mixed with sterile physiological saline (0.9%) and the turbidity was adjusted to the standard inoculum of Mac-Farland scale 0.5 (106 colony forming units (CFU) per ml). Petriplates containing 20 ml of Mueller Hinton Agar was used for antibacterial activity. The inoculum was spread on the surface of the solidified media and Whatman No. 1 filter paper discs (5 mm in diameter) impregnated with the test compound (20 μl/disc) were placed on the plates.
Ampicillin (10 mg/disc) was used as positive control for bacteria. A paper disc impregnated with petroleum ether was used as a negative control. Plates inoculated with the bacteria were incubated for 24 hour at 37°C. The inhibition zone diameters were measured in centimeters. All the tests were performed in triplicate, The E test (AB Biodisk) which is a quantitative method for antimicrobial susceptibility testing was applied and the standard deviation has been measured by the conventional measure of repeatability and the average was taken as final reading [33-37].
Results and Discussion
The copper soap and complex are abbreviated as follows-
- Copper - Neem Soap (CN)
- Copper – Neem Soap Urea Complex (CNU)
IR spectral analysis
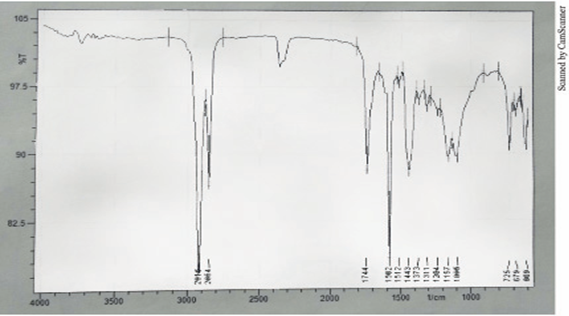
The absorption bands observed at 2925 cm–1 and 2854 cm–1 corresponds to asymmetric and symmetric stretching of methylene (-CH2) group. The presence of absorption bands at 1458 cm––1 is representative of symmetric bending of nearly 3010 cm–1 corresponds to olefinic =C–H stretch. The strong absorption band at 1665 cm–1 and another weaker band at 1373 cm–1 were due to carboxylate ion COO–, C–O anti symmetric and symmetric stretching respectively. Also >C=O stretching bands were observed at 1744 cm–1 small peak corresponding to –CH2 twisting and wagging has been observed at 1311 cm–1. Also peaks corresponding to –CH3 and –CH2 rocking have been seen at 1157 cm–1 and 725 cm–1 respectively. Copper–oxygen (Cu–O) stretching bands have been distinguished at 480 cm–1 [23-25].
The absorption bands observed at 2925 cm–1 and 2854 cm–1 corresponds to asymmetric and symmetric stretching of methylene (-CH2) group. The presence of absorption bands at 1458 cm––1 is representative of symmetric bending of nearly 3010 cm–1 corresponds to olefinic =C–H stretch. The strong absorption band at 1665 cm–1 and another weaker band at 1373 cm–1 were due to carboxylate ion COO–, C–O anti symmetric and symmetric stretching respectively. Also >C=O stretching bands were observed at 1744 cm–1 small peak corresponding to –CH2 twisting and wagging has been observed at 1311 cm–1. Also peaks corresponding to –CH3 and –CH2 rocking have been seen at 1157 cm–1 and 725 cm–1 respectively. Copper–oxygen (Cu–O) stretching bands have been distinguished at 480 cm–1 [23-25].
| Absorption bands | CN (cm-1) | CNU (cm-1) |
| Corresponding to soap moiety | ||
| Olefinic =C–H stretching | 3010 | 3009 |
| CH3 and CH2, C–H Anti sym. stretching (vas) | 2916 | 2925 |
| CH3 and CH2, C–H sym. stretching (vs) | 2854 | 2854 |
| > C = O Stretching | 1744 | 1744 |
| C = C stretching [Cis Unconjugated] | 1665 | - |
| COO -, C–O Anti-sym. Stretching | 1582 | 1589 |
| CH2, C–H Bending (δ) (Scissoring) | 1465 | 1458 |
| C–H, Deformation, =C–H Rocking | 1443 | - |
| COO–, C–O Sym. Stretching | 1373 | 1373 |
| CH2, C–H Bending (δ) (Twisting and Wagging) | 1311 | 1311 |
| CH3, C–H Rocking | 1157 | - |
| CH2, C–H Rocking | 725 | 765 |
| =C-H, Out of Plane Bending of C-H | 679 | 705 |
| Cu–O Stretching | 480 | 560 |
| Corresponding to ligand moiety | ||
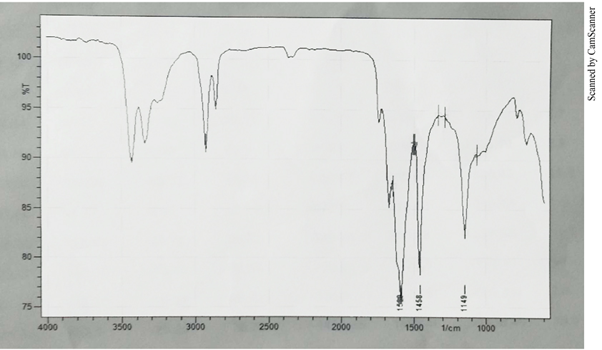
| NH2, N–H Stretching | - | 3500 |
| Sym. NH2, N–H Stretching | - | 3420 |
| N-H Bending | - | 1651 |
| C–N Stretching | - | 1149 |
Table 3: IR spectral data for copper (II) Neem Soap and its complex.
The above-mentioned absorption bands (Table 3) were found to be common with the absorption bands observed for pure copper soap of non-edible oils. Apart from these absorption bands the following bands were also observed corresponding to the ligand moiety [38-40]. The C–N stretching band of primary amide was observed at nearly 1149 cm–1 the absorption band 1744 cm–1 was found to be representative of amide > C = O group. A broad band near 3500-3420 cm–1 was observed corresponding to N–H stretching of amides. Also C–H stretching band due to deformation out of plane (in Benzene) was also observed at 705 cm–1 (Table 3) [41-44]. Thus on the basis of above observations it can be safely assumed that complexation of copper soap has taken place with urea.
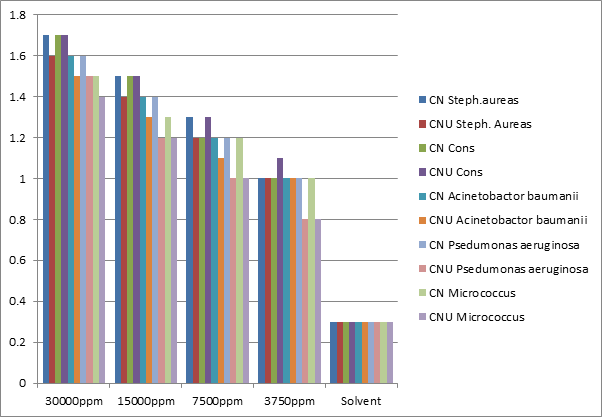
| Concertration (PPM) | CN S.aureus (cm) | CNU S. aureus (cm) | CN Cons(cm) | CNU Cons(cm) | CN A. baumanii(cm) | CNU A. baumanii (cm) | CN P. aeruginosa (cm) | CNU P. aeruginosa (cm) | CNMicrococcus (cm) | CNU Micrococcus(cm) |
| 3 * 104 | 1.7 | 1.6 | 1.7 | 1.7 | 1.6 | 1.5 | 1.6 | 1.5 | 1.5 | 1.4 |
| 1.5 * 104 | 1.5 | 1.4 | 1.5 | 1.5 | 1.4 | 1.3 | 1.4 | 1.2 | 1.3 | 1.2 |
| .75 * 104 | 1.3 | 1.2 | 1.2 | 1.3 | 1.2 | 1.1 | 1.2 | 1 | 1.2 | 1 |
| .3750 * 104 | 1 | 1 | 1 | 1.1 | 1 | 1 | 1 | 0.8 | 1 | 0.8 |
| Solvent | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
Table 4: Bactericidal data for copper (II) soap and its complex derived from Neem oil.
Biological Activities
Neem urea soap complex show higher antibacterial activity than pure soap suggesting that complex is more powerful antibacterial agent and other N & S etc. containing compounds are able to enhance the performance of copper soap. The enhanced activity of newly synthesized complex as compared to the soap can possibly be explained on the basis of presence of donor atoms N and O as well as the structural compatibility with molecular nature of the toxic moiety [34,45]. The activity of copper Neem soap and copper Neem soap urea complex derived from Neem oil were found in the follow order soap and complex respectively:
Neem urea soap complex show higher antibacterial activity than pure soap suggesting that complex is more powerful antibacterial agent and other N & S etc. containing compounds are able to enhance the performance of copper soap. The enhanced activity of newly synthesized complex as compared to the soap can possibly be explained on the basis of presence of donor atoms N and O as well as the structural compatibility with molecular nature of the toxic moiety [34,45]. The activity of copper Neem soap and copper Neem soap urea complex derived from Neem oil were found in the follow order soap and complex respectively:
For Soap
Staphylococcus aureus= Coagulase-negative staphylococci (CoNs) > Acinetobactor baumanii = Pseudomonas aeruginosa > Micrococcus
Staphylococcus aureus= Coagulase-negative staphylococci (CoNs) > Acinetobactor baumanii = Pseudomonas aeruginosa > Micrococcus
For Complex
Coagulase-negative staphylococci (CoNs) > Staphylococcus aureus > Acinetobactor baumanii > Pseudomonas aeruginosa > Micrococcus
Coagulase-negative staphylococci (CoNs) > Staphylococcus aureus > Acinetobactor baumanii > Pseudomonas aeruginosa > Micrococcus
Conclusion
Antiseptic soap, sometimes called antibacterial soap or anti-fungal soap, is a regular soap in liquid or solid form. Though these compounds are considered to have low toxicity, their 2-hydroxy isomers have been shown to undergo thermal and photo-chemical ring closure to form polychlorinated dibenzo-p-dioxins which are highly toxic. In addition, it is though that bacteria could develop resistance to triclosa and this could lead to development of resistance and change in microbial community structure. These shortcomings of antiseptic soap (treated with synthetic antiseptics) generate the need to develop more environment friendly toilet soaps [46-48].
Neem oil was particularly chosen as it is easily available commercially and biodegradable in nature. Copper soap have a tendency of complexation with ‘nitrogen’ and ‘oxygen’ containing ligands. The interest in co-ordination chemistry is increasing continuously with the preparation of organic ligands containing a variety of donor groups and it is multiplied many folds when the ligand have biological importance. These facts led us to synthesize copper soaps of non-edible oils and its complex with ligand containing nitrogen and oxygen elements, investigate the role of copper and other elements in biological activities [49-50]. The compounds studied are comparatively better antibacterial agents than Imipenem [IPM10] which showed lower antibacterial activity.
Acknowledgement
The authors are thankful to UGC for JRF and Principal and Head of Department (SPC Govt. College, Ajmer) for laboratory facilities. We are also thankful to Dr. P.K. Khatri (Head of Department Microbiology, Dr. S.N. Medical, Jodhpur) Dr. Archana Bora and Dr. R.S. Parihar for their kind cooperation.
The authors are thankful to UGC for JRF and Principal and Head of Department (SPC Govt. College, Ajmer) for laboratory facilities. We are also thankful to Dr. P.K. Khatri (Head of Department Microbiology, Dr. S.N. Medical, Jodhpur) Dr. Archana Bora and Dr. R.S. Parihar for their kind cooperation.
References
- Khan S., et al. “Antifungal Activities of Copper Surfactants derived from Neem (AzadirectaIndica) and Karanj (Pongamiapinnata) Oils: A Pharmaceutical Application”. Global Journal of Pharmacy & Pharmaceutical Sciences 3.4 (2017): 1-6.
- R Sharma and S Khan. “RPMP Natural Products”. 18 (2007): 429.
- MRK Sherwani., et al. “Micellar features and other solution properties of copper (II) soaps in benzene”.Indian Journal of Chemistry42A (2003): 2527-2530.
- N Singh., et al. Pest Management. Sci 56 (2000): 28.
- GN Mukherjee and A Das. “Borate complexes in solution. Part-II. Complex formation equilibria of some cis-diaqua-Co-III complexes with boric acid”. Journal of the Indian Chemical Society 78.2 (2001): 78-81.
- KN Mehrotra., et al. Cellulose Chemistry and Technology 1 (1973): 38.
- Swamy SJ and Reddy SR. “Synthesis and characterization of manganese (II), cobalt (II), nickel (II) copper (II) and zinc (II) complexes of new polydentate ligands containing 1, 8-naphthyridine moiety”. Indian Journal of Chemistry40(A) (2001): 1093.
- Siegel SH and Martin RB. Chemical Reviews 82 (1983): 235.
- MRK Sherwani., et al. “Micellar features and other solution properties of copper (II) soaps in benzene”.Indian Journal of Chemistry42A (2003): 2527-2530.
- Mehta VP and Sharma R. Tenside Surfactants Detergents 40.2 (2003): 99.
- May PM and Bulman RA. “5 The Present Status of Chelating Agents in Medicine”. Progress in Medicinal Chemistry 20 (1983): 225-336.
- Dutta RL and Syamal A. Elements of Magnetochemistry 2nd Ed Sci India Sect A65 (1995): 247.
- Ryan KJ., et al. “Sherris medical microbiology an introduction to infectious diseases” 4th ed McGraw-Hill New York (2004):
- Salyers AA and Whitt DD. “Bacterial pathogenesis: a molecular approach, Second Edition”. ASM Press (2002):
- Kloos WE and Bannerman TL. “Update on clinical significance of coagulase-negative staphylococci”. Clinical Microbiology Reviews 7.1 (1994): 117–140.
- Pfaller MA and Herwaldt LA. “Laboratory, clinical, and epidemiological aspects of coagulase-negative staphylococci”. Clinical Microbiology Reviews1.3 (1988): 281–299.
- Taponen S and Pyörälä S. “Coagulase-negative staphylococci as cause of bovine mastitis—not so different from Staphylococcus aureus?” Veterinary Microbiology 134.1-2 (2009): 29–36.
- Brown RW. “Composition of scientific words: a manual of methods and a lexicon of materials for the practice of logotechnics”. Rev. ed. Smithsonian Institution Press, Washington D.C (1956):
- Larson E., et al. “Physiologic microbiologic and seasonal effects of hand washing of the skin of health care personal”. 14 (1986): 51.
- William Riley. “In Topley & Wilson s Microbiology & Microbial infections”. 2 (2005): 1301-1305.
- Arun K Singh and Raj K Singh. “A Search for Eco-friendly Detergent /Dispersant Additives for Vegetable-Oil Based Lubricants”. Journal of Surfactants and Detergents15.4 (2012): 399–409.
- Joseph J and Boomadevi JG. “Synthesis, structural characterization and biological studies of copper complexes with 2-amino benzothiazole derivatives”. Journal of Molecular Structure 1063 (2014): 160-169.
- Sinkou H., et al. “Video – Observed treatment for tuberculiosis patients in Belarus: Findings from the first Programmatic experience”. European Respiratory Society49.3 (2017): 1602049.
- Silver S. “Membrane and Transport” 2.115 (1982): 7.
- Ojha KG., et al. Indian Chemical Society 79 (2002): 191.
- Sharma VK. and Shrivastava S. “Synthesis and characterization of trivalent chromium, manganese, iron and cobalt complexes with Schiff bases derived from 4-amino-5-mercapto-l, 2, 4-triazoles”. Indian Journal of Chemistry45A (2006): 1368-1374.
- Basset J., et al. “Vogels Textbook of Quantitative Inorganic Analysis”. Longman, London (1985):
- Riddick J.R. and Bunger W.B. “Organic Solvents Physical Properties and Methods of Purification”. Wiley Interscience New York (1970):
- Gunstone FD. “An Introduction to the Chemistry of Fats and Fatty Acids”. Chapman and Hall Ltd, London (1958).
- Adams R. “Organic Reactions”. Wiley (1959): 3240.
- Mehta VP., et al. “Tenside Surfactants Detergents”. HANSER eLibrary16 (1979): 2.
- Manhas BS., et al. Journal of the Indian Chemical Society 83 (2006): 652.
- Reddy RK., et al. “Synthesis, characterization, antibacterial and anthelmentic activities of copper (II) complexes with benzofuran Schiff bases”. Indian Journal of Chemistry 45A (2006): 377.
- AK Sharma., et al. IJMSET 2.12 (2015): 31-40.
- TV Sivanandan. “Minister: India Close to Setting U Petrochemicals Complex in Iran”. The Hindu (2016):
- DA Kalashnikov., et al. “Accessing photon bunching with a photon number resolving multi-pixel detector”. Optics Express 19.10 (2011): 9352.
- Cassina M., et al. “First-trimester exposure to metformin and risk of birth defects: A systematic review and meta-analysis”.Human Reproduction Update20.5 (2014): 656-669.
- Feldsine P., et al. “AOAC International methods committee guidelines for validation of qualitative and quantitative food microbiological official methods of analysis”. Journal of AOAC INTERNATIONAL85.5 (2002): 1187–1200.
- DJMac Lowry., et al. “Journal of Applied Microbiology 20 (1970): 46-53.
- Portillo R Vila., et al. “Antifungal activity of Paraguayan plants used in traditional medicine”. Journal of Ethnopharmacology76.1 (2001): 93-98.
- Silverstein., et al. “Spectrometric Identification of Organic Compounds”. Journal of Mass Spectrometry (1991): 430.
- Bansod NH., Chaudhari, G.N., Thihale, M.S. and Patil, S.D. Sie Pharma Chhemica 5.2 (2013): 144-148.
- Lal RA., et al. “Binuclear and tetranuclear dioxouranium (VI), zinc (II) and copper (II) complexes derived from bis (2-hydroxy -1-naphthaldehyde) malonoyldihydrazone”. Indian Journal of Chemistry45A (2006): 619.
- Patel RN., et al. “Synthetic, spectroscopic and electrochemical behaviour of biomimetic copper (II) complex with CuN4 chromophore”. Indian Journal of Chemistry 45A (2006): 614.
- Mijanuddin M., et al. “Synthesis, spectroscopic and crystallographic studies on two copper (II) benzimidazole complexes: Supramolecular frameworks built from N-H ... O, N-H ... Cl and O-H ... Cl hydrogen bonds”. Indian Journal of Chemistry45A (2006): 858.
- Ali A., et al. “Mono- and di-nuclear Cu (II) complexes of p-tert-butyl-calix [4]arene-1,3-diacid , derivative: A comparative study of their characterization and catecholase mimetic activity”. Indian Journal of Chemistry 45A (2006): 853.
- Erden I., et al. “Synthesis and characterization of a new Schiff base ligand derived from benzo-15- crown-5 and its complexes with cobalt (II), nickel (II) and copper (II)”. Indian Journal of Chemistry 45A (2006): 1395-1399.
- BA Arthington-Skaggs., et al. “Comparative Evaluation of PASCO and National Committee for Clinical Laboratory Standards M27-A Broth Micro dilution Methods for Antifungal Drug Susceptibility Testing of Yeasts”. Journal of Clinical Microbiology 38.6 (2000): 2254-2260.
- L Rocha A., et al. Antibacterial phloroglucinols and flavonoids from Hypericum brasiliense”. Phytochemistry 40.5 (1995): 1447-1452.
- Tank P., et al. “A Pharmaceutical approach & Antifungal activities of Copper Soaps with their N & S donor complexes derived from Mustard and Soyabean oils”. Global Journal of Pharmacy & Pharmaceutical Sciences 3.4 (2017): 1-6.
Citation:
Priyanka Kumawat., et al. “Synthesis and Characterization of Bactericidal Agents Derived from Neem (Azadirecta Indica) Oil”.
Clinical Biotechnology and Microbiology 1.5 (2017): 209-218.
Copyright: © 2017 Priyanka Kumawat., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.