Case Report
Volume 1 Issue 1 - 2017
Unique Presentation of Pulmonary Metastasis Nine Years Following Resection of Primary Renal Cell Carcinoma
1Resident Physician, Internal Medicine, PinnacleHealth Hospitals, Harrisburg, PA, U.S
2Research Director, Associate Program Director, Internal Medicine Residency, PinnacleHealth Hospitals, Harrisburg, PA, U.S
3Medical Director, PinnacleHealth Robotics Institute, Cardiovascular and Thoracic Surgery, PinnacleHealth Hospitals, Harrisburg, PA, U.S
2Research Director, Associate Program Director, Internal Medicine Residency, PinnacleHealth Hospitals, Harrisburg, PA, U.S
3Medical Director, PinnacleHealth Robotics Institute, Cardiovascular and Thoracic Surgery, PinnacleHealth Hospitals, Harrisburg, PA, U.S
*Corresponding Author: Dr. Saketram Komanduri, Resident Physician, Internal Medicine, PinnacleHealth Hospitals, Harrisburg, PA,
U.S.
Received: February 17, 2017; Published: April 08, 2017
Abstract
Metastatic disease after resection of localized renal cell carcinoma (RCC) is not uncommon. The lung is the most common site of
metastasis from renal cell carcinoma, usually occurring within three years of primary resection. Most individuals are asymptomatic,
with few patients presenting with symptoms such as chest pain or discomfort. This case is significant in that the patient had experienced
symptoms of severe chest tightness over the course of six months, initially misconstrued as cardiac in origin, and eventually
discovered to be due to a metastatic lung lesion eroding into the adjacent chest wall. The patient’s presentation had been unique with
regards to time of occurrence as well, as disease free interval (DFI) had been almost ten years. In view of this anomalous presentation
and time of occurrence, the patient was subject to an extensive cardiac work-up due to suspicion of cardiac disease rather than the
potential presence of a metastatic lesion, placing the patient at risk for malignant chest wall infiltration had diagnosis been delayed.
Introduction
Renal cell carcinomas (RCC) account for approximately 80-85% of primary renal neoplasms. Metastatic disease following resection
of primary RCC is not uncommon. Approximately 30% of cases are metastatic either at the time of diagnosis or during follow-up [1]. The
ability of RCC to metastasize to even uncommon sites is due to its rich vasculature and lymphatic drainage [2]. The most common site of
metastasis is the lungs. Most patients with pulmonary metastatic disease are asymptomatic at diagnosis [3-5]. Chest wall metastasis is
rare, with only a few cases of metastasis via lymphatic or vascular pathways reported.
Disease free interval (DFI) varies, with the risk of recurrence greatest in the first two to three years following resection of RCC. The
case in point exemplifies a unique presentation of pulmonary metastases potentially involving the interior chest wall, following a disease
free interval of almost ten years.
Case Presentation
A 73 year old male had repeatedly presented to primary care and cardiology clinics with complaints of chest pain and discomfort for the past six months. The patient’s extensive cardiac history was significant for multiple coronary interventions, including four vessel CABG in 2006 and a recent abnormal stress test. Concern for a cardiac etiology of his symptoms prompted an extensive cardiac workup. A dedicated CT of the chest during the patient’s work-up revealed the presence of a 1.2 cm left upper lobe lung nodule. Prior to this finding, the patient had been diagnosed with primary clear cell renal cell carcinoma nine years ago, treated with resection, and had been disease free since then. Initial pathology and staging of the primary renal cell tumor was pT1b, NX, MX; Fuhrman nuclear grade 2 with negative margins. The patient’s past medical history is otherwise significant for hypertension, hyperlipidemia, chronic obstructive pulmonary disease and obesity.
A 73 year old male had repeatedly presented to primary care and cardiology clinics with complaints of chest pain and discomfort for the past six months. The patient’s extensive cardiac history was significant for multiple coronary interventions, including four vessel CABG in 2006 and a recent abnormal stress test. Concern for a cardiac etiology of his symptoms prompted an extensive cardiac workup. A dedicated CT of the chest during the patient’s work-up revealed the presence of a 1.2 cm left upper lobe lung nodule. Prior to this finding, the patient had been diagnosed with primary clear cell renal cell carcinoma nine years ago, treated with resection, and had been disease free since then. Initial pathology and staging of the primary renal cell tumor was pT1b, NX, MX; Fuhrman nuclear grade 2 with negative margins. The patient’s past medical history is otherwise significant for hypertension, hyperlipidemia, chronic obstructive pulmonary disease and obesity.
Cardiothoracic Surgery and Interventional Radiology were consulted leading to a PET scan and biopsy of the nodule. Biopsy was
positive for clear cell type of RCC and a PET scan was otherwise negative for additional metastatic disease. Left video-assisted thoracoscopic
surgery (VATS) wedge resection, pleural biopsy, and lymph node dissection was subsequently performed. During the removal of
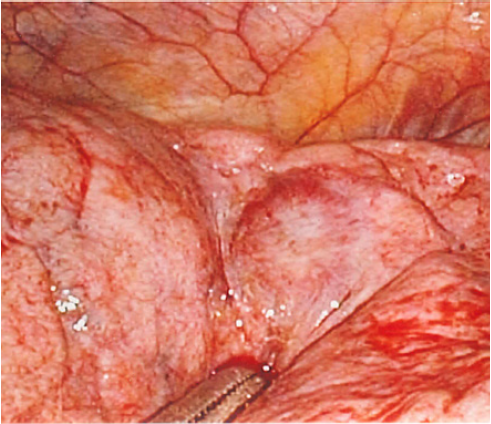
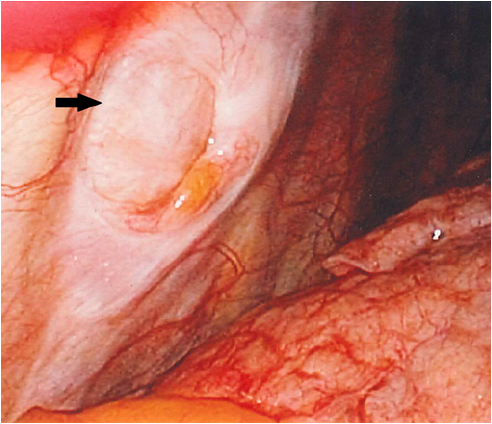
the metastatic nodule, a defect was noted in the adjacent chest wall concerning for erosion and possible metastatic spread. The mass was
not adherent to the chest wall; however a defect was present in the intercostal muscles where the mass had been (see Figures 1 and 2).
A decision was made to biopsy the eroded chest wall area and to perform chest wall reconstruction if the biopsy proved to be positive
for metastatic disease. The area of the defect was biopsied and sent for pathology. Wedge excision was negative at the resection margin,
pleural surface, and for metastatic disease to the chest wall. The patient’s post-operative course was uneventful.
Discussion
RCC is known for its tendency to metastasize well after curative resection of the primary tumor. Risk of recurrence is highest in the
first two to three years, with emphasis on surveillance for at least five years. Beyond five years, continuation of surveillance is based on
a case-to-case basis.
As per guidelines compiled by the American Urological Association (AUA) and the National Comprehensive Cancer Network (NCCN),
surveillance is comprised of history and physical examination, basic laboratory studies, and chest and abdominal imaging with either CT
or radiograph. Additional imaging is based on clinical suspicion [6,7]. It has been debated that stopping surveillance after five years has
been associated with missed recurrences, and that extending protocol to fifteen years post-nephrectomy would detect approximately
95% of recurrences, with an obvious undesired increase in cost as well [8]. Similar to the case in point, many reports have shown that
recurrences occur well beyond the five-year mark [9], portraying the importance of maintaining a high index of suspicion in patients
with a history of RCC.
Patients presenting with pulmonary metastasis are usually asymptomatic, with a few exceptions. The patient mentioned in this case
presented with symptoms due to a unique presentation, in which the nodule was eroding into the adjacent chest wall. Chest pain or
discomfort is a non-specific symptom with a vast differential diagnosis. Given the patient’s extensive cardiac history, his chest pain was
easily misconstrued as being of cardiac origin. Metastases usually involve lymphatic and vascular pathways, especially in the case of RCC,
owing to the kidney’s rich vascular supply. Chest wall metastasis has been described to anterior or posterior ribs; however no case has
been reported involving spread to the intercostal muscles of the chest wall adjacent to the visceral pleura. As with all cancers, contiguous
spread due to direct infiltration of tumor cells (such as that involved in the spread of RCC through the Gerota’s fascia and into the adrenal
gland) is always a possibility. The patient had a clear erosion of the chest wall, and although the metastatic nodule was not adherent to
the wall, direct malignant infiltration into the chest wall was a potential possibility.
Progression free survival is an important end-point in treatment of patients with malignancy. In patients with lung metastasis,
metastatectomy is a potential curative approach [10]. Good prognostic factors for pulmonary metastases include the presence of solitary
metastasis as well as small number and small size (< 3 cm) of metastases. Favorable outcomes are linked to metachronous rather than
synchronous metastasis, and a disease-free interval greater than three years between initial nephrectomy and subsequent diagnosis of
pulmonary metastases carries a better prognosis [9,11]. The majority of these apply to the case in point.
Conclusion
Patients presenting with non-specific complaints with a history of malignancy should be assessed and investigated with a high index
of suspicion, regardless of DFI. Erosion into the adjacent chest wall is a unique presentation of pulmonary metastases, which exemplifies
the potential of other distinctive metastatic pathways. Regardless of the surveillance plan, it is imperative that clinicians, both specialists
and internists, deal with patients with a history of malignancy using a broad approach, thus avoiding telescopic views leading to delayed
diagnostic or therapeutic interventions.
References
- Seigel R., et al. “Cancer Statistics”. CA: A Cancer Journal for Clinicians65.1 (2015): 5-29.
- Staderini F., et al. “A unique presentation of renal clear cell carcinoma with atypical metastases”. International Journal of Surgery Case Reports11 (2015): 29-32.
- Ljungberg B., et al. “Follow-up guidelines for nonmetastatic renal cell carcinoma based on the occurence of metastases after radical nephrectomy”. BJU International 84.4 (1999): 405-411.
- Levy D., et al. “Stage specific guidelines for surveillance after radical nephrectomy for local renal cell carcinoma”. Journal of Urology 159.4 (1998):1163-1167.
- Hafez K., et al. “Patterns of tumor recurrence and guidelines for followup after nephron sparing surgery for sporadic renal cell carcinoma”. Journal of Urology 157.6 (1997): 2067-2070.
- Motzer R., et al. “Kidney cancer, version 3”. Journal of the National Comprehensive Cancer Network 13 (2015): 151-159.
- Donat S., et al. “Follow-up for clinically localized renal neoplasms: AUA Guideline”. Journal of Urology 190.2 (2013): 407-416.
- Stewart S., et al. “Evaluation of the National Comprehensive Cancer Network and American Urological Association renal cell carcinoma surveillance guidelines”. Journal of Clinical Oncology 32.36 (2014): 4059-4065.
- Tsakiridis K., et al. “Lost in time pulmonary metastases of renal cell carcinoma: complete surgical resection of metachronous metastases, 18 and 15 years after nephrectomy”. Journal of Thoracic Disease (Suppl 1) (2012): 69-73.
- Pfannschmidt J., et al. “Surgical intervention for pulmonary metastases”. Deutsches Ärzteblatt International 109.40 (2012): 645-651.
- Sponholz S., et al. “Results of Pulmonary Resection: Other Epithelial Malignancies”. Thoracic Surgery Clinics 26.1 (2016): 99-108.
Citation:
Dr. Saketram Komanduri., et al. “Unique Presentation of Pulmonary Metastasis Nine Years following resection of Primary
Renal Cell Carcinoma”. Pulmonary Research and Respiratory Care 1.1 (2017): 20-23.
Copyright: © 2017 Dr. Saketram Komanduri., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.