Research Article
Volume 1 Issue 4 - 2017
Presenting Patterns of Retinoblastoma at Sekuru Kaguvi Eye Hospital in Harare, Zimbabwe
Department of Ophthalmology, University of Zimbabwe College of Health Sciences, P. O. Box A178, Avondale. Harare. Zimbabwe
*Corresponding Author: Madzima P, Department of Ophthalmology, University of Zimbabwe College of Health Sciences, P. O. Box A178, Avondale. Harare. Zimbabwe.
Received: October 24, 2017; Published: November 08, 2017
Abstract
Background: Retinoblastoma is the most common primary intraocular malignancy of childhood world over and Zimbabwe has the third highest incidence worldwide. Despite the high incidence, published literature on retinoblastoma in Zimbabwe is limited.
Objectives: To characterise the presenting patterns of retinoblastoma amongst patients attending Sekuru Kaguvi Eye Hospital (SKH), determine the recurrence rate, mortality rate and factors associated with mortality amongst these patients.
Study design: Hospital based retrospective records review and patient recall.
Methodology: A retrospective cross-sectional study in which medical records of patients admitted at SKH from 1st January 2010 to 31st December 2014 with a histological diagnosis of retinoblastoma were reviewed. Those fitting the inclusion criteria were recalled back to the hospital where they had an ocular examination and computed tomography (CT) scan of the orbits and brain.
Results: Eighty-seven patients had a histological diagnosis and fitted the inclusion criteria. The mean age was 32.8 months (median 27 months). There was no gender predilection and two thirds (58/87) of the cases were unilateral. Proptosis was the most common presenting symptom accounting for 64.4% of cases (56/87). The mean paternal age was 31.8 years (median 29 years). Over 60% of patients (59/87) had traditional eye medications (TEM) used on them. Amongst the 68 patients that were followed up, cases of death as reported by the guardians were 61.8% (42/68). Orbital recurrence amongst survivors was 28%. Mortality in patients with retinoblastoma at SKH was significantly associated with older age at presentation, female gender, bilateral disease, presenting with proptosis and use of TEM.
Conclusions: Proptosis, late presentation with advanced disease, high mortality rate and unacceptably high loss to follow-up are the characteristics of RB seen among admitted patients at Sekuru Kaguvi Eye Hospital in Zimbabwe.
Keywords: Retinoblastoma; Proptosis; Leucocoria; Childhood; Cancer
Abbreviations: SKH: Sekuru Kaguvi Eye Hospital; CT: Computed tomography; TEM: Traditional eye medicines; RB: Retinoblastoma; USA: United States of America; DRC: Democratic Republic of Congo
Introduction
Retinoblastoma (RB) is the most common primary intraocular malignancy of childhood occurring in about 1 in 17 000 live births. [1] Zimbabwe is estimated to have the third highest incidence worldwide. [2] This malignancy accounts for about 3% of all childhood cancers worldwide but in Zimbabwe this proportion is doubled. [1,3] Despite the high incidence in Zimbabwe, published literature on RB in this country is limited.
In developed countries, leukocoria is the most common presenting symptom of RB accounting for about 60% of cases followed by strabismus (20%). [1] In contrast, a pilot study carried out at SKH in 2013 reported that proptosis was the most common presenting symptom accounting for 62% of cases followed by leukocoria (29%). [4] Advanced paternal age has been reported to be associated with RB [1] however, the above mentioned pilot study at SKH reported a mean paternal age of 31 years. An audit conducted in 2013 reported that RB was the most common cause of deaths amongst paediatric admissions at SKH. [5] Although this condition is curable orbital recurrence of RB following enucleation is about 4.2% in the United States of America (USA) as reported by Kim., et al. [6] Unfortunately, literature on the recurrence of RB in Africa is limited [6] and the recurrence at SKH is unknown. All these observations prompted for further research into RB to be conducted at SKH.
Materials and Methods
Study design
This was a retrospective cross-sectional hospital based study in which medical records of patients admitted at SKH between 1 January 2010 and 31 December 2014 with a diagnosis of RB were reviewed and patients recalled for assessent.
This was a retrospective cross-sectional hospital based study in which medical records of patients admitted at SKH between 1 January 2010 and 31 December 2014 with a diagnosis of RB were reviewed and patients recalled for assessent.
Study setting
This study was conducted at SKH, the largest tertiary eye institute at Parirenyatwa Group of Hospitals in Zimbabwe.
This study was conducted at SKH, the largest tertiary eye institute at Parirenyatwa Group of Hospitals in Zimbabwe.
Study population
All patients who presented and were admitted to SKH with an eye condition during the period from 1 January 2010 to 31 December 2014. This time period was selected because the country’s economy was stabilising following introduction of multicurrency system making medical care more accessible to the general public.
All patients who presented and were admitted to SKH with an eye condition during the period from 1 January 2010 to 31 December 2014. This time period was selected because the country’s economy was stabilising following introduction of multicurrency system making medical care more accessible to the general public.
Inclusion criteria
- Patients admitted to SKH during the period under review with a diagnosis of RB.
- Patients whose notes were available.
- Patients who had a histological diagnosis of RB.
Exclusion criteria
- Patients with RB who were re-admitted at SKH during the period under review.
- Patients whose notes were not available.
- Those who did not have a histological diagnosis of RB.
Those fitting the criteria were recalled back to the hospital where they had an ocular examination and computed tomography (CT) scan of the orbits and brain.
Data processing and analysis
Data was analysed using Epi-Info version 7.1.4. The characteristics of the study subjects were evaluated using descriptive statistics. Univariate analysis was carried out to identify factors associated with mortality in RB. Results were evaluated at a 95% confidence interval and at a significance level of p < 0.05.
Data was analysed using Epi-Info version 7.1.4. The characteristics of the study subjects were evaluated using descriptive statistics. Univariate analysis was carried out to identify factors associated with mortality in RB. Results were evaluated at a 95% confidence interval and at a significance level of p < 0.05.
Results
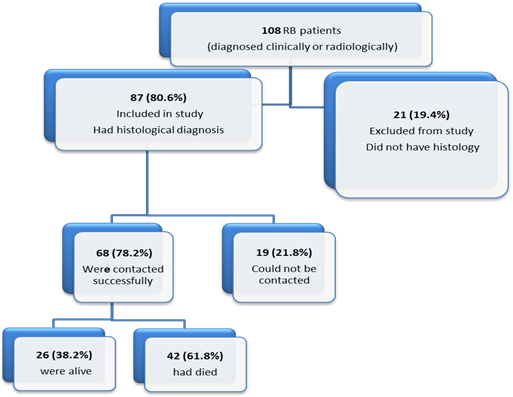
A total of 108 patients were admitted at SKH over the 5 year period with a clinical, radiological or histological diagnosis of RB. Of these, 87 fitted the inclusion criteria as shown in Figure 1 below.
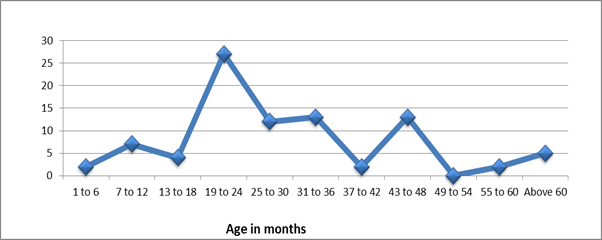
Patients’ age at presentation ranged from 1 month to 156 months (13 years) with mean age of 32.8 months and median of 27 months. (Figure 2 below)
Gender distribution: There was no gender predilection. The male to female ratio was 1:1.1. X2 test, p = 0.621.
Laterality: About two thirds (66.7%) of the study population had unilateral disease.
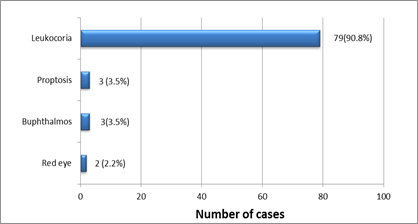
Earliest symptom noticed: Leukocoria was the earliest symptom noticed by caregivers in 90.8% of cases (79/87) as shown in Figure 3 below.
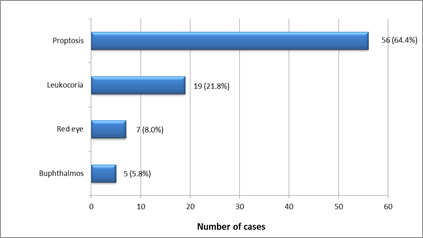
Presenting symptom: The majority of patients (64.4%; 56/87) presented with proptosis as shown in figure 4 below.
Delay in presenting to hospital: The period of delay in presenting to hospital was that from the day the first symptom was noticed by the caregiver to the day the patient presented to SKH. Patients delayed by a period that ranged from 3 weeks to 4 years. The average period was 10 months.
Delay in treatment: The period from radiological diagnosis to treatment ranged from 1 day to 90 days and the average period of delay in commencing treatment was 10 days.
Paternal Age: Paternal age ranged from 22 years to 50 years with mean of 31.8 years and median of 29 years.
Traditional Eye Medicine use: Fifty-nine of the 87 cases (67.8%) had information regarding TEM use. Forty of them (67.8%) had guardians who admitted to using TEM on the child. The other 19 (32.2%) denied use.
Recurrence of retinoblastoma: In this study, orbital recurrence was defined as evidence of orbital tumour on CT scan amongst patients who had tumour confined to the globe on histology following enucleation. Of the 26 surviving patients, 25 of them had tumour confined to the globe on histology following enucleation and 1 had optic nerve involvement. Seven of the 25 patients (28%) with intraocular tumour had orbital recurrence.
Mortality: At the time of follow-up, cases of death as reported by the parent or guardian were 61.8% see figure 1.
Factors associated with mortality
- Older age of patient: Patients who died were older (median age 29 months) than those who survived (median age 24 months). X2 test, p = 0.018
- Female gender: Females were 1.5 times less likely to survive compared to males. X2 test, 95% CI = 0.92 – 2.46; RR = 1.50; p = 0.044
- Bilateral disease: A larger proportion of bilateral cases (90.9%) died compared to unilateral cases (56.5%). X2 test, p = 0.001
- Presenting with proptosis: Majority of patients who presented with proptosis (72.1%) died compared to those without proptosis (44%). X2 test, p = 0.010
- TEM use: TEM users were 1.4 times less likely to survive compared to non-users. X2 test, 95% CI = 0.90 – 2.26; RR = 1.43; p = 0.045
Discussion
Age at presentation
Patients with RB in developing countries are known to present to hospital much later than those in developed countries. The overall mean age at presentation in this study was 32.8 months. Comparing with a study by Dr Tumushime conducted here between 1987 and 1988 where the mean age was 30 months, the mean age at presentation of patients with RB at SKH has remained unchanged over the past 25 years. [7] Affected patients in South Africa, Kenya and DRC have a similar mean age at presentation. [8-10] In Mali, RB patients present at much older ages and has the highest mean age at presentation of 50 months. [11] Patients in the USA and the UK present much earlier and mean age at presentation is about 18 months. [12]
Patients with RB in developing countries are known to present to hospital much later than those in developed countries. The overall mean age at presentation in this study was 32.8 months. Comparing with a study by Dr Tumushime conducted here between 1987 and 1988 where the mean age was 30 months, the mean age at presentation of patients with RB at SKH has remained unchanged over the past 25 years. [7] Affected patients in South Africa, Kenya and DRC have a similar mean age at presentation. [8-10] In Mali, RB patients present at much older ages and has the highest mean age at presentation of 50 months. [11] Patients in the USA and the UK present much earlier and mean age at presentation is about 18 months. [12]
The age distribution in our study peaked at 24 months and 48 months. The second peak could indicate that more of the older children who were at home with the disease were beginning to present to hospital, suggesting improved awareness.
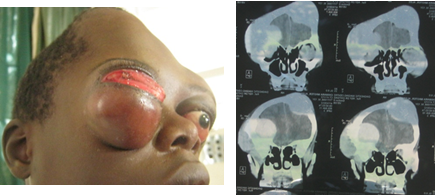
Figure 5: 13 year old, late presenter with bilateral RB, skull and intracranial metastases confirmed on imaging.
Presenting Symptoms
In developed countries, leukocoria is the most common presenting symptom of RB accounting for about 60% of cases followed by strabismus (20%). [1] The most common presenting symptom in our study was proptosis accounting for nearly two- thirds (64.4%) of cases followed by leukocoria which accounted for about a fifth of cases (21.8%). Other developing countries show similar results with proptosis being the commonest presenting symptom. [11, 13-15] The predominance of proptosis at presentation is due to late presentation. [16] South Africa and DRC however, are different with most of their patients presenting with leukocoria. [17,10] Twenty-five years ago, leukocoria used to be the most frequent presenting symptom of RB amongst patients in Zimbabwe [7] but now they present with proptosis. The change in pattern of presentation documented here could be attributed to economic challenges being experienced that result in limited access to medical care in Zimbabwe.
In developed countries, leukocoria is the most common presenting symptom of RB accounting for about 60% of cases followed by strabismus (20%). [1] The most common presenting symptom in our study was proptosis accounting for nearly two- thirds (64.4%) of cases followed by leukocoria which accounted for about a fifth of cases (21.8%). Other developing countries show similar results with proptosis being the commonest presenting symptom. [11, 13-15] The predominance of proptosis at presentation is due to late presentation. [16] South Africa and DRC however, are different with most of their patients presenting with leukocoria. [17,10] Twenty-five years ago, leukocoria used to be the most frequent presenting symptom of RB amongst patients in Zimbabwe [7] but now they present with proptosis. The change in pattern of presentation documented here could be attributed to economic challenges being experienced that result in limited access to medical care in Zimbabwe.
Late Presentation
On average, patients in this study delayed to present to hospital by a period of about 10 months. This period is shorter in South Africa (7 months), DRC (6.5 months) and Iran (7.4 months) where the majority of patients present with leukocoria. [9,10,18] Developed countries have even shorter delays ranging from 2 to 3 months. [19-21] In our study, although the majority of caregivers had noticed leukocoria in the patient, they took the child to a health facility after proptosis had appeared. Reasons for presenting late included seeking traditional medication, late referral from primary health centres, lack of finances to travel and some guardians who suspected cataract preferred to have the child operated at an older age and so delayed the hospital visit. About two thirds of the study population at SKH used TEM and they were 1.5 times more likely to present with proptosis compared to non-users. Patients delayed to present to hospital because of visiting traditional healers, using TEM and often, presented when there was no improvement. It was observed that guardians often sought medical attention for proptosis which was more disfiguring and painful than leukocoria.
On average, patients in this study delayed to present to hospital by a period of about 10 months. This period is shorter in South Africa (7 months), DRC (6.5 months) and Iran (7.4 months) where the majority of patients present with leukocoria. [9,10,18] Developed countries have even shorter delays ranging from 2 to 3 months. [19-21] In our study, although the majority of caregivers had noticed leukocoria in the patient, they took the child to a health facility after proptosis had appeared. Reasons for presenting late included seeking traditional medication, late referral from primary health centres, lack of finances to travel and some guardians who suspected cataract preferred to have the child operated at an older age and so delayed the hospital visit. About two thirds of the study population at SKH used TEM and they were 1.5 times more likely to present with proptosis compared to non-users. Patients delayed to present to hospital because of visiting traditional healers, using TEM and often, presented when there was no improvement. It was observed that guardians often sought medical attention for proptosis which was more disfiguring and painful than leukocoria.
Paternal age
An association between aging and mutations in germ line cells has been demonstrated, [22] suggesting a higher incidence of childhood malignancies (including RB) among children of older parents. [23] Matsunanga., et al. in Japan reported an increased risk of RB in children with fathers aged 35 years and older and Dockerty., et al. in the UK reported that fathers aged 45 years and older were 3 times more likely to have children with RB compared to those who were younger. [24,25] The mean paternal age in countries such as Iran, Japan, Netherlands and France ranges from 32.3 years to 34.4 years. [18,24,26,27] Literature on similar studies in Africa is limited. The mean paternal age in our study population was 31.8 years suggesting that advanced paternal age may not be a risk factor for the development of RB in patients presenting to SKH.
An association between aging and mutations in germ line cells has been demonstrated, [22] suggesting a higher incidence of childhood malignancies (including RB) among children of older parents. [23] Matsunanga., et al. in Japan reported an increased risk of RB in children with fathers aged 35 years and older and Dockerty., et al. in the UK reported that fathers aged 45 years and older were 3 times more likely to have children with RB compared to those who were younger. [24,25] The mean paternal age in countries such as Iran, Japan, Netherlands and France ranges from 32.3 years to 34.4 years. [18,24,26,27] Literature on similar studies in Africa is limited. The mean paternal age in our study population was 31.8 years suggesting that advanced paternal age may not be a risk factor for the development of RB in patients presenting to SKH.
Gender distribution
There was a slight female predominance in this study with male to female ratio of 1: 1.1 however this was not statistically significant. Several studies from different parts of the world also report no gender predilection. [10,12,28,29,30] There are however other studies conducted in Mali, Kenya, DRC, and China that reported male predominance. [8,11,31] Nyamori., et al. reported that in Kenya, the male predominance suggested that caregivers preferentially sought medical treatment for the boy child over the girl child however no studies have been conducted to prove that assumption. [8]
There was a slight female predominance in this study with male to female ratio of 1: 1.1 however this was not statistically significant. Several studies from different parts of the world also report no gender predilection. [10,12,28,29,30] There are however other studies conducted in Mali, Kenya, DRC, and China that reported male predominance. [8,11,31] Nyamori., et al. reported that in Kenya, the male predominance suggested that caregivers preferentially sought medical treatment for the boy child over the girl child however no studies have been conducted to prove that assumption. [8]
Laterality
About two thirds of the RB cases in this study were unilateral. These findings are consistent with those in developed countries but differ with other populations. In Mali about 90% of cases are unilateral and studies conducted at two hospitals in Nigeria observed no bilateral cases. [11,32,33] In the Asian population of Britain, more than three quarters of RB cases are unilateral. [34]
About two thirds of the RB cases in this study were unilateral. These findings are consistent with those in developed countries but differ with other populations. In Mali about 90% of cases are unilateral and studies conducted at two hospitals in Nigeria observed no bilateral cases. [11,32,33] In the Asian population of Britain, more than three quarters of RB cases are unilateral. [34]
Recurrence of retinoblastoma
Orbital recurrence in this study was 28%. The high orbital recurrence could have been a consequence of treatment delay, defaulting treatment and poor surgical technique. Amongst in-patients at SKH the period from diagnosis to treatment ranged from 1 day to 90 days (mean 10 days) therefore some patients could have already had microscopic extraocular spread of the tumour by the time the enucleation was performed. Patients who defaulted treatment had an even greater risk of orbital recurrence. SKH is a teaching hospital and the majority of enucleation surgeries are performed by doctors in training who could be practising poor surgical techniques that result in tumour seeding. On the other hand, the high orbital recurrence could have been an indication of chemotherapy failure especially in those that developed orbital recurrence even after completing the full treatment regimen without defaulting. Chan., et al. reported that failure of chemotherapy is associated with expression of the multidrug resistance protein p170 which frequently occur in RB. Perhaps some of our patients who developed recurrence expressed this protein. Cyclosporine inhibits p170 making chemotherapy failures rare [35] however cyclosporine is not routinely used in the treatment of patients who present to SKH because of availability challenges.
Orbital recurrence in this study was 28%. The high orbital recurrence could have been a consequence of treatment delay, defaulting treatment and poor surgical technique. Amongst in-patients at SKH the period from diagnosis to treatment ranged from 1 day to 90 days (mean 10 days) therefore some patients could have already had microscopic extraocular spread of the tumour by the time the enucleation was performed. Patients who defaulted treatment had an even greater risk of orbital recurrence. SKH is a teaching hospital and the majority of enucleation surgeries are performed by doctors in training who could be practising poor surgical techniques that result in tumour seeding. On the other hand, the high orbital recurrence could have been an indication of chemotherapy failure especially in those that developed orbital recurrence even after completing the full treatment regimen without defaulting. Chan., et al. reported that failure of chemotherapy is associated with expression of the multidrug resistance protein p170 which frequently occur in RB. Perhaps some of our patients who developed recurrence expressed this protein. Cyclosporine inhibits p170 making chemotherapy failures rare [35] however cyclosporine is not routinely used in the treatment of patients who present to SKH because of availability challenges.
Mortality in patients with retinoblastoma
The mortality rate of RB in developing countries is estimated to be 40%. [36] Mortality rates as high as 90% have been reported in DRC. [37] In our study, cases of death as reported by the parent or guardian were 61.8%. The mortality rate could not be accurately calculated because the date of death could not be ascertained for some patients and death certificates could not be obtained. It was therefore uncertain whether RB was the cause of death.
The mortality rate of RB in developing countries is estimated to be 40%. [36] Mortality rates as high as 90% have been reported in DRC. [37] In our study, cases of death as reported by the parent or guardian were 61.8%. The mortality rate could not be accurately calculated because the date of death could not be ascertained for some patients and death certificates could not be obtained. It was therefore uncertain whether RB was the cause of death.
The most important factor associated with high mortality was late presentation as suggested by Chantada., et al. [38] This was observed in our study as patients who presented at an older age and with more advanced disease (proptosis or bilateral disease) were less likely to survive. Developed countries have very low mortality rates due to early detection and appropriate treatment. Some of these countries perform genetic testing which can predict children at high risk of developing RB in utero. [39]
More females died compared to males and this could have been because there were slightly more females in the study or it was an indication of preferential treatment of the boy child over the girl child in the study population supporting the hypothesis by Nyamori., et al. in Kenya. [8]
TEM use was found to be associated with high mortality. TEM users were 1.5 times more likely to present with proptosis and were 1.4 times less likely to survive compared to non-users. Those who used TEM delayed coming to hospital and often presented when the condition had not improved with TEM. As a result, they were more likely to present with advanced disease associated with poorer prognosis.
Conclusion
Retinoblastoma is characterised by proptosis being the leading presenting symptom, late presentation with advanced disease, high mortality rate and unacceptably high loss to follow-up rate at Sekuru Kaguvi Eye Hospital in Zimbabwe.
Recommendations
- Awareness campaigns and screening programs for retinoblastoma should be implemented and these could be linked to immunisation programs. Social media could be used as an awareness tool and baby photographs could be screened for the red reflex with the use of smart phone applications.
- A registry for children with retinoblastoma should be created, containing all contact details so as to improve follow-up. A loss to follow-up rate of 21.8% is too high to ignore when we expect quality service provision.
- Treatment should be made more accessible by decentralizing treatment facilities and a toll free line should be provided so that guardians with affected children get prompt expert advice over the phone.
Acknowledgements
We acknowledge the role played by all members of staff at Sekuru Kaguvi Eye Hospital in assisting us retrieve patient information from records.
We acknowledge the role played by all members of staff at Sekuru Kaguvi Eye Hospital in assisting us retrieve patient information from records.
Conflict of interest
This study was not sponsored by any organization and there is no conflict of interest.
This study was not sponsored by any organization and there is no conflict of interest.
References
- Kanski J and Browling B. “Clinical Ophthalmology: a systematic approach. 7th edition”. Edinburgh: Elsevier Saunders(2009): 510-513.
- Parkin DM., et al. “International incidence of childhood cancer”. International Journal of Cancer 42.4 (1988): 511-520.
- Chokunonga E., et al. “Cancer incidence in Harare Triennal report: 2010-2012”.
- Madzima P. “A retrospective study to establish the incidence, presentation, management and survival rate of retinoblastoma patients at a tertiary eye institute in Zimbabwe”. Thesis submitted to the University of Zimbabwe in partial fulfilment of Masters in Medicine Ophthalmology Degree (2014):
- Madzima P and Masanganise R. “Five-year mortality rate in an ophthalmic ward in Zimbabwe”. African Vision and Eye Health 74.1 (2015): 4.
- Kim JW., et al. “Orbital recurrence of retinoblastoma following enucleation”. British Journal of Ophthalmology 93.4 (2009): 463-467.
- Tumushime BF. “Management of retinoblastoma at Sekuru Kaguvi Eye Unit, Parirenyatwa hospital, Harare, Zimbabwe”. Submitted to Department of Surgery Faculty of Medicine, University of Zimbabwe in June 1995 in partial fulfilment of Masters in Medicine Ophthalmology Degree.
- Nyamori JM., et al. “The incidence and distribution of retinoblastoma in Kenya. British Journal of Ophthalmology96.1 (2012): 141-143.
- Aim´e Kazadi Lukusa., et al. “Retinoblastoma in the Democratic Republic of Congo: 20-Year Review from a Tertiary Hospital in Kinshasa”. Journal of Cancer Epidemiology 2012.920468 (2012): 5.
- Goolam S., et al. “Retinoblastoma in South Africa - A 20-year retrospective study at two tertiary academic hospitals in Johannesburg”. Investigative Ophthalmology & Visual Science 56 (2015):
- Boubacar T., et al. “A 30 month prospective study on the treatment of retinoblastoma in the Gabriel Toure Teaching Hospital, Bamako, Mali”. British Journal of Ophthalmology94.4 (2010): 467-469.
- Broaddus E., et al. “Incidence of retinoblastoma in the USA: 1975-2004”. British Journal of Ophthalmology93.1 (2009): 21-23.
- Saiju R., et al. “Retinoblastoma in Nepal: a clinical profile of 30 cases”. Nepal Medical College Journal 8.3 (2006): 171-175.
- Ozdemir H., et al.“Clinical and epidemiological characteristics of retinoblastoma: correlation with prognosis in a Turkish Pediatric Oncology Center”. Pediatric Hematology and Oncology24.3 (2007): 221-231.
- Poso MY., et al. “Malignant tumours of the eye and adnexa in Congo-Kinshasa”. Journal Francais d’Ophtalmologie 23.4 (2000): 327-332.
- Antoneli CB., et al. “The pediatrician’s ability to recognize the presenting signs and symptoms of retinoblastoma”. Revista da Associac¸˜ao M´edica Brasileira50.4 (2004): 400-402.
- Akang EE., et al. “Retinoblastomas in Ibadan Nigeria-II: Clinicopathologic features”. West African Journal of Medicine 19.1 (2000): 6-11.
- Nabie R., et al. “Characteristics and clinical presentations of paediatric retinoblastoma in North-Western Iran”. International Journal of Ophthalmology5.4 (2012): 510-512.
- Goddard A., et al. “Delay in diagnosis of retinoblastoma: risk factors and treatment outcome”. British Journal of Ophthalmology83.12 (1999): 1320-1323.
- Jensen RD and Miller RW. “Retinoblastoma epidemiologic characteristics”. The New England Journal of Medicine285 (1971): 307-311.
- Wirix M., et al. “Delayed diagnosis of retinoblastoma”. Bulletin De La Societe Belge D'Ophtalmologie 278 (2000): 37-41.
- Mills MB., et al. “Mutation risk associated with paternal and maternal age in a cohort of retinoblastoma survivors”. Human Genetics131.7 (2011): 1115-1122.
- Yip BH., et al. “Parental age and risk of childhood cancers: A population-based cohort study from Sweden”. International Journal of Epidemiology35.6 (2006): 1495-1503.
- Matsunaga E., et al. “Parental age and seasonal variation in the births of children with sporadic retinoblastoma: A mutation-epidemiologic study”. Human Genetics 84.2 (1990): 155-158.
- Dockerty JD., et al. “Case–Control study of parental age, parity and socioeconomic level in relation to childhood cancers”. International Journal of Epidemiology 30.6 (2001): 1428-1437.
- DerKinderen DJ., et al. “Parental age in sporadic hereditary retinoblastoma”. American Journal of Ophthalmology110.6 (1990): 605-609.
- Pellié C., et al.“Parental age in retinoblastoma”. Humangenetik 20.1 (1973): 59-62.
- Lea-Leal C., et al. “A multicentre report from the Mexican retinoblastoma group”. British Journal of Ophthalmology88.8 (2004): 1074-1077.
- MacCarthy A., et al. “Retinoblastoma in Great Britain 1963-2002”. British Journal of Ophthalmology93.1 (2009): 33-37.
- Arif M., et al. “Retinoblastoma in NWFP, Pakistan”. Journal of Ayub Medical College Abbottabad21.4 (2009): 60-62.
- Bai S., et al. “Retinoblastoma in the Beijing Tongren Hospital from 1957 to 2006: clinicopathological findings”. British Journal of Ophthalmology95.8 (2010): 1072-1076.
- Obafunwa JO., et al. “Pediatric malignancies in Plateau state, Nigeria”. The Cancer Journal 5 (1995): 211-215.
- Tijani SO., et al. “Morphological patterns of pediatric solid cancer in Lagos, Nigeria”. West African Journal of Medicine14.3 (1995): 174-180.
- Stiller CA., et al. “Childhood cancer an ethnic group in Britain: A United Kingdom children cancer study group”. British Journal of Cancer64.3 (1991): 543-548.
- Chan HS., et al. “Multidrug resistance protein (MRP) expression in retinoblastoma correlates with the rare failure of chemotherapy despite cyclosporine for reversal of P-glycoprotein”. Cancer Research57.12 (1997): 2325-2330.
- Kivela T. “The epidemiological challenge of the most frequent eye cancer: Retinoblastoma, an issue of birth and death”. British Journal of Ophthalmology 93.9 (2009): 1129-1131.
- Kaimbo Wa Kaimbo D., et al.“Presenting signs of retinoblastoma in Congolese Patients”. Bulletin De La Societe Belge D'Ophtalmologie283 (2002): 37-41.
- Chantada GL., et al. “Strategies to manage retinoblastoma in developing countries”. Pediatric Blood & Cancer56.3 (2011): 341-348.
- Gallie BL., et al. “Global issues and opportunities for optimized retinoblastoma care”. Pediatric Blood & Cancer49.7 (2007): 1083-1090.
Citation:
Madzima P and Masanganise R. “Presenting Patterns of Retinoblastoma at Sekuru Kaguvi Eye Hospital in Harare, Zimbabwe”.
Ophthalmology and Vision Science 1.4 (2017): 149-157.
Copyright: © 2017 Madzima P and Masanganise R. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.



































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.