Editorial
Volume 2 Issue 3 - 2018
Innate Immunotherapy of Recurring Glioblastomas: Preliminary Trials with Neutrophils
International Institute of Medicine and Science, California, USA
*Corresponding Author: Alain L Fymat, International Institute of Medicine and Science, California, USA.
Received: April 04, 2018; Published: April 27, 2018
Abbreviations: BBB: Blood Brain Barrier; GB: Glioblastomas; GBM: Glioblastomas Multiform; NDD: Neuro Degenerative Disorder; PTX: Paclitaxel; TMZ: Temozolomide
Drugs cited: Paclitaxel; Temozolomide.
Glioblastoma (GB) or Glioblastoma multiform (GBM), the most common primary brain tumor in adults, remains an unmet need in oncology. It is the most aggressive and most common brain cancer, the second most common after meningioma, representing 15% of brain tumors. Its annual incidence is ~ 3.19 cases per 100,000 population. Several risk factors have been identified, including: genetic disorders (increased incidence of gliomas; neurofibromatosis types 1 and 2; tuberous sclerosis); certain syndromes (Von Hippel-Lindau; Li Fraumeni; Turcot) or diseases (astrocytoma); medical exposure (previous radiation therapy); occupational exposure (lead); ethnicity (Caucasians; Hispanics; Asians); gender (slightly more common in men than women); and age (over 50, most commonly around 64 years of age). However, no prevention strategy is known. While first-line treatment has been clearly defined since 2005, no standard second-line treatment has yet been determined. The survival rate is ~ 1 year, and only 5% of the people affected survive for 5 years.
The standard treatment consists of: (a) surgery (maximal resection) followed by (b) radio chemotherapy (6 weeks of radiotherapy at a dose of 60 Grey [Gy]) together with concomitant chemotherapy with Temozolomide (TMZ) at a rate of 75 mg/m2 daily) (TMZ is an alkalyting agent used as a first-line treatment for GB and as a second-line treatment for astrocytomas) and, once radio chemotherapy is complete, (c) adjuvant treatment (a minimum of 6 months with TMZ is started at a dose of 150–200 [mg/m2] for 5 days every 28 days). Nonetheless, most treatments cannot eradicate all tumor cells. Several of the available treatment strategies employed either singly or in combination have been reviewed earlier [1-3], including: surgery, chemotherapy, conformal radiotherapy, boron neutron therapy, intensity modulated proton beam therapy, antiangiogenic therapy, alternating electric fields, vaccines, palliative therapies and even lifestyle changes. Unfortunately, surgery is often insufficient given the diffuse nature of the disease. Even when tumors have been surgically removed, deeply infiltrated cancer cells often remain and contribute to relapse; chemotherapy has major limitations because most drugs cannot cross the blood brain barrier (BBB), and penetration into brain cells is limited. In addition, the cells in brain tumors are greatly heterogeneous, which limits the treatment efficacy and explains the high rate of progression of the disease.
More recently, in a study utilizing three mouse models of human GBM, Xue., et al. at the China Pharmaceutical University have devised a neutrophil-mediated anticancer nanotechnological drug delivery for the suppression of postoperative malignant glioma recurrence [4]. Neutrophils are white blood cells in the granulocytic series of blood cell development (see Figure 1 illustrating the several steps a blood stem cell goes through to become a red blood cell, a platelet, or a white blood cell). Formed by myelopoietic tissue of the bone marrow and released into the circulating blood, where they normally represent 54-65% of the total number of leukocytes, they penetrate inflamed brain tumours. Prior animal and human studies had reported that neutrophils can cross the BBB, and although they are not typically attracted to GBMs, they are recruited at sites where tumors had been removed in response to post-operative inflammation. Taking advantage of the characteristics of these innate immune cells, Xue and his research team manufactured liposome capsules that encased Paclitaxel (PTX), a traditional chemotherapy drug, with lipids, loaded them into neutrophils and injected them in the blood of three mouse models of GBM after surgical resection.
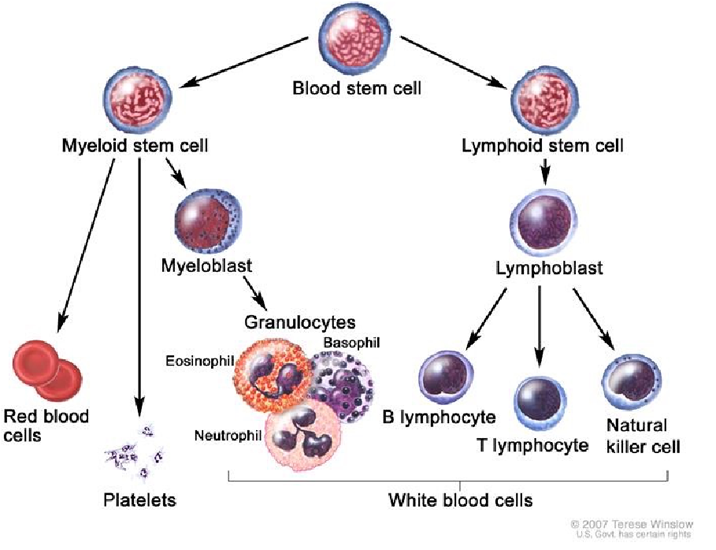
Figure 1: Illustrating neutrophils in blood cell development.
(Source: National Cancer Institute Archives)
(Source: National Cancer Institute Archives)
When the treatment was applied following surgical removal of the main tumor mass, the neutrophil-carrying drugs were able to penetrate the BBB, destroy residual cancer cells, and slow the growth of new tumors. Inflammatory factors released after tumour resection guide the movement of the neutrophils into the inflamed brain. The highly concentrated inflammatory signals in the brain trigger the release of liposomal PTX from the neutrophils, allowing delivery of PTX into the remaining invading tumour cells. This neutrophil-mediated delivery of drugs efficiently slowed the recurrent growth of tumours, significantly improved survival rates, but did not completely inhibit the regrowth of tumours. While tumor recurrence was not completely prevented, overall, mice receiving this treatment lived significantly longer than controls.
One strength of the above method is, as stated earlier, that neutrophils are the most abundant white blood cells, so they can be collected in significant amount from a patient's blood. However, there are at least three limitations of the method used: (1) It used approximately ten times the number of neutrophils found in normal mouse circulation. Thus, the blood amount needed for this type of procedure in humans could be quite substantial; (2) Two of the mouse models used are disliked by the neuro-oncology community because they elicit an immune rejection response; and (3) the third model is based on a human GBM cell line that is different from the original tumor source. Nonetheless, although additional studies are necessary to further validate the method, the strategy of using neutrophils to deliver drugs across the BBB could be applied to human GBMs, neurodegenerative diseases (NDDs), other inflammation-mediated disorders, and any other diseases that naturally attract neutrophils.
References
- Fymat AL. “Glioblastoma therapies: Where do we stand?” Med Plus Journal of Cancer and Oncology Research 1.1 (2017): 1-12.
- Fymat AL (2017). “Surgical and non-surgical management and treatment of glioblastoma: I. Primary tumors”. Open Access Journal of Surgery 7.2 (2017):1-8.
- Fymat AL. “Surgical and non-surgical management and treatment of glioblastoma: II. Recurring Tumors”. Open Access Journal of Surgery 7.1 (2017): 1-7.
- Xue J., et al. “Neutrophil-mediated anticancer drug delivery for suppression of postoperative malignant glioma recurrence”. Nature Nanotechnology12 (2017): 692–700.
Citation:
Alain L Fymat. “Innate Immunotherapy of Recurring Glioblastomas: Preliminary Trials with Neutrophils”. Current Opinions in
Neurological Science 2.3 (2018): 480-482.
Copyright: © 2018 Alain L Fymat. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.






























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.