Editorial
Volume 1 Issue 1 - 2018
A Rationale for using Gabapentinoids in Topical Analgesic Formulations?
Professor of molecular pharmacology, pain specialist Institute for Neuropathic Pain, Bosch en Duin, The Netherlands
*Corresponding Author: Jan M Keppel Hesselink, Professor of molecular pharmacology, pain specialist
Institute for Neuropathic Pain, Bosch en Duin, The Netherlands.
Received: November 02, 2017; Published: February 24, 2018
In a Cochrane analysis in 2017, topical analgesic medication was defined as a medication applied to body surfaces such as the skin or mucous membranes to treat pain, where the formulation is either rubbed onto the skin or made into patches or plasters that are stuck onto the skin. [1] So, what analgesics are most fit for use in topical analgesic formulations? A simple question, and as we will demonstrate, not an easy one to answer. More and more topical therapies are regarded as safe, well-tolerated, and are an effective alternative to oral analgesics in the treatment of chronic musculoskeletal and neuropathic pain. [2] The question, ‘What analgesics are most fit for using in topical analgesic formulations?’ is therefore a key one in this field. We will discuss this issue related to a topical formulation containing gabapentin.
In a recent paper on the topical treatment of skin pain, it was pointed out that there was a large body of evidence available for the use of gabapentin and pregabalin in the treatment of chronic pain state. [3] According to the author, it was therefore unsurprising that physicians used gabapentin and pregabalin compounded in topical formulations to treat neuropathic pain, more so because the existing oral formulations can easily be crushed.
This line of thought is relevant, as it demonstrates the structure and level of argumentation leading to the selection of an analgesic in a topical formulation against pain. Apparently only two reasons are being put forward: 1. the compound needs to be a proven, established analgesic, and 2. one must be able to crush the formulation into a powder. The latter argument is of course only relevant for the local compounding pharmacist, and is not much of a structural requirement as one can obtain pure compound via different providers. The first argument therefore seems the more important one: the active pharmaceutical ingredient needs to be an established analgesic. Now, this implies that all established analgesics would be fit to be formulated into a topical application.
Let us discuss one key issue in selecting an analgesic for a topical formulation to treat neuropathic pain: the site of action of the active pharmaceutical ingredient (e.g. intra-dermal versus transdermal or systemically). After this discussion, we will be better equipped to answer the question whether gabapentinoids are useful in topical formulations.
The site of action of the active pharmaceutical ingredient related to transdermal and dermal delivery systems
The indicator ‘topical’ is rarely defined, because most authors apparently feel it is obvious. However, the term can refer to the place where the formulation is applied (localized area of the skin), but it can also refer to the site where the active ingredient has its mechanism of action.
The indicator ‘topical’ is rarely defined, because most authors apparently feel it is obvious. However, the term can refer to the place where the formulation is applied (localized area of the skin), but it can also refer to the site where the active ingredient has its mechanism of action.
In the first case, all formulations applied to the skin are topical: creams, gels, plasters, foams, etc. For instance, both capsaicin and lidocaine plasters, as well as a PLO amitriptyline gel would qualify as a topical analgesic formulation. This would not be very helpful for our thinking. Capsaicin stays locally and exerts its action via the effect at local epidermal targets (the TRPV-1 channel and the nociceptors). Amitriptyline in PLO gel however, is designed to lead to adequate plasma levels in the range of oral amitriptyline, and its mechanism of action is therefore a central one. [4] PLO gel is a transdermal delivery system for the active pharmaceutical ingredient, comparable to fentanyl plasters. While a capsaicin plaster is aimed at bringing a high dose of capsaicin directly into the epidermis to create a neuropathological effect, the dying back of small fibers, a fentanyl plaster is designed to create a sufficient steady state in plasma for systemic analgesia.
A differentiation of topical analgesics according to the main mechanism of action is more telling: in the compartments of the skin (dermal delivery systems), or systematically, as in transdermal delivery systems. This directly implies that the application of a transdermal delivery system needs to result in sufficient plasma levels of the active ingredient, while this is not the case for dermal delivery systems (see Figure 1).
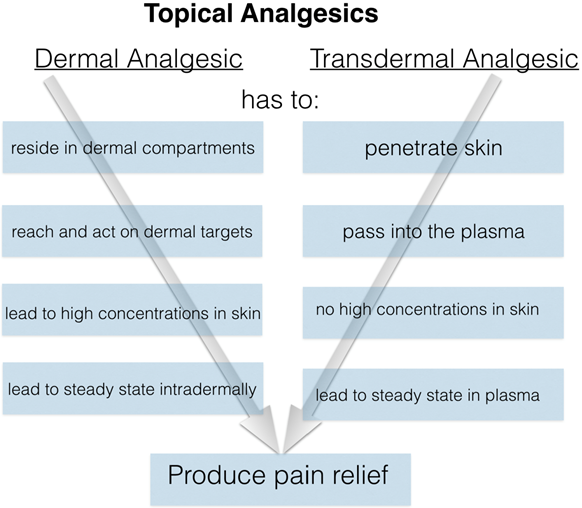
Figure 1: Both dermal and transdermal analgesics are based on topical formulations.
A dermal analgesic formulation has its mechanism of action residing in the skin, while
a transdermal delivery system containing an analgesic, is designed to deliver sufficient
of this active pharmaceutical ingredient to the plasma.
If we add gabapentin or pregabalin to a topical cream, we aim to create either sufficient plasma levels in the blood (in the case of a transdermal delivery system), or we avoid such levels as in a dermal delivery system. In the latter case, after application we will be able to detect only very low concentrations in the blood as the formulation is aimed to reach high concentrations in the epidermal tissues only. Now in case of gabapentin, the aim is to treat pain ‘residing in the skin’, as Scheinfeld in his paper on topical analgesics for skin-pain pointed out [3]. Aiming at such a dermal formulation indeed seems often to be the case, and urine concentrations of topical gabapentin are much lower compared to oral formulations.
Glinn., et al. in 2017 published the results of data from over 29,000 specimens of patients treated with topical analgesics; he tested for urine concentrations of gabapentin, ketamine, cyclobenzaprine or amitriptyline, either administered orally or topically. [5] In only 12% of topically treated cases, gabapentin was above the level of detection. Of these positive cases, most patients had gabapentin levels present below 1500 ng/mL. In patients treated orally, most patients had levels far higher at > 10000 ng/mL.
Thus, it seems the aim of a topical gabapentin formulation is to reach high drug levels in the skin, while avoiding high plasma levels. This implies that the mechanism of action of dermal delivery systems containing gabapentin should be residing in the skin. That however is not the case, as we will find out.
A mechanism of action of topical gabapentin?
Gabapentin was initially designed as a drug that needed to be able to cross the blood/brain barrier and interact with central GABAergic systems. Later it appeared that its target was not the GABAergic system, but the α2-δ subunit of voltage gated calcium channels. This channel is now regarded as the main target of gabapentin; after its specific binding to this subunit analgesia occurs. In most reviews its pain attenuating effect is after oral intake, explained via its acting on both the central nervous system (on the spinal and the supra-spinal areas) as well as on the peripheral region, at the level of the dorsal root ganglion (DRG) neurons and dorsal horn neurons [6]. This α2-δ protein subunit of the voltage-gated calcium channels is the high, nano-molar-affinity binding site for gabapentinoids, and to date no other high-affinity molecular targets have been described. [7] The indications of epilepsy and anxiety disorders for gabapentin also are pointers for targets in the central nervous system. Its indication of neuropathic pain leans on both central as well as peripheral targets (DRG and dorsal horn neurons). If gabapentin applied topically (in a transdermal delivery system) does not result in sufficient plasma levels, the compound will not reach its targets. Interestingly, we have not come across any systematic measurements of gabapentin in the plasma after topical application. Only the findings in the urine are available and these indicate low systemic levels, definitely under the levels that would be required for obtaining systemic effects.
Gabapentin was initially designed as a drug that needed to be able to cross the blood/brain barrier and interact with central GABAergic systems. Later it appeared that its target was not the GABAergic system, but the α2-δ subunit of voltage gated calcium channels. This channel is now regarded as the main target of gabapentin; after its specific binding to this subunit analgesia occurs. In most reviews its pain attenuating effect is after oral intake, explained via its acting on both the central nervous system (on the spinal and the supra-spinal areas) as well as on the peripheral region, at the level of the dorsal root ganglion (DRG) neurons and dorsal horn neurons [6]. This α2-δ protein subunit of the voltage-gated calcium channels is the high, nano-molar-affinity binding site for gabapentinoids, and to date no other high-affinity molecular targets have been described. [7] The indications of epilepsy and anxiety disorders for gabapentin also are pointers for targets in the central nervous system. Its indication of neuropathic pain leans on both central as well as peripheral targets (DRG and dorsal horn neurons). If gabapentin applied topically (in a transdermal delivery system) does not result in sufficient plasma levels, the compound will not reach its targets. Interestingly, we have not come across any systematic measurements of gabapentin in the plasma after topical application. Only the findings in the urine are available and these indicate low systemic levels, definitely under the levels that would be required for obtaining systemic effects.
Our experience with gabapentin 10% and phenytoin 10%
Some 8 years ago we started developing topical formulations containing a variety of (co-)analgesics, such as baclofen, amitriptyline, ketamine, clonidine and gabapentin. For gabapentin at that time we selected a fairly high concentration, 10%, related to the fact that its potency compared to pregabalin was relatively low. While most patients were quite satisfied with most of our compounded creams, we felt that the gabapentin cream had disappointing results. We subsequently selected a different anticonvulsant, phenytoin, and meanwhile we treated more than 80 patients with the latter, resulting in more satisfied patients. We also assessed plasma levels in 16 patients who were treated with our topical phenytoin cream, and in all of the cases the plasma level was not above the limit of detection. From the perspective presented above the discrepancy is clear: gabapentin has no targets in the skin, while phenytoin is a broad acting sodium channel blocker and has multiple targets in the epidermal: the cross-talking triad consisting of nociceptors, keratinocytes and immune-modulatory cells (see Figure 2).
Some 8 years ago we started developing topical formulations containing a variety of (co-)analgesics, such as baclofen, amitriptyline, ketamine, clonidine and gabapentin. For gabapentin at that time we selected a fairly high concentration, 10%, related to the fact that its potency compared to pregabalin was relatively low. While most patients were quite satisfied with most of our compounded creams, we felt that the gabapentin cream had disappointing results. We subsequently selected a different anticonvulsant, phenytoin, and meanwhile we treated more than 80 patients with the latter, resulting in more satisfied patients. We also assessed plasma levels in 16 patients who were treated with our topical phenytoin cream, and in all of the cases the plasma level was not above the limit of detection. From the perspective presented above the discrepancy is clear: gabapentin has no targets in the skin, while phenytoin is a broad acting sodium channel blocker and has multiple targets in the epidermal: the cross-talking triad consisting of nociceptors, keratinocytes and immune-modulatory cells (see Figure 2).
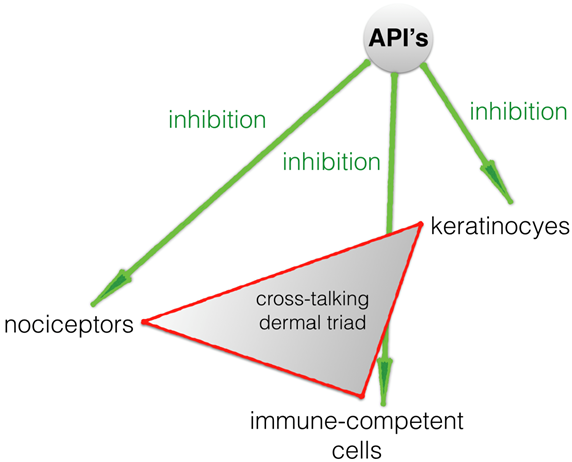
Figure 2: The targets for dermal formulation containing the API phenytoin: sodium channels on the cross-talking triad in the epidermal tissue: nociceptors, keratinocytes and immune-modulatory cells. Phenytoin is a broad sodium channel blocker and can inhibit overactive cells in the epidermis, resulting in a reduction of pain in localized neuropathic pain. Other APIs with comparable targets are also suited for topical dermal formulations, such as lidocaine. For gabapentin no dermal targets are known, making it unlikely that a dermal gabapentin formulation would be effective in reducing localized neuropathic pain.
The answer: analgesics with dermal targets
This leads us to the answer to the question, ‘What analgesics are most fit for using in topical analgesic formulations?’ There are many requirements for such analgesics, but at least the targets should reside in the skin. Phenytoin has such targets, while gabapentinoids like gabapentin have not. Gabapentin and related molecules therefore are only fit for transdermal formulations, and such formulations should result in sufficient plasma levels of the API in question. Dermal formulation will not result in sufficient plasma levels of gabapentinoids and thus will remain inactive.
This leads us to the answer to the question, ‘What analgesics are most fit for using in topical analgesic formulations?’ There are many requirements for such analgesics, but at least the targets should reside in the skin. Phenytoin has such targets, while gabapentinoids like gabapentin have not. Gabapentin and related molecules therefore are only fit for transdermal formulations, and such formulations should result in sufficient plasma levels of the API in question. Dermal formulation will not result in sufficient plasma levels of gabapentinoids and thus will remain inactive.
Conflict of interest
The author is a patent holder of two patents related to the topical formulations of phenytoin in the treatment of neuropathic pain.
The author is a patent holder of two patents related to the topical formulations of phenytoin in the treatment of neuropathic pain.
References
- Derry S., et al. “Topical analgesics for acute and chronic pain in adults - an overview of Cochrane Reviews”. The Cochrane Database of Systematic Reviews 12.5 (2017).
- Stanos SP and Galluzzi KE. “Topical therapies in the management of chronic pain”. Postgrad Med 125(4 Suppl 1) 2013: 25-33.
- Scheinfeld N. “Topical treatments of skin pain: a general review with a focus on hidradenitis suppurativa with topical agents”. Dermatology Online Journal 20.7 (2014).
- Scott MA., et al. "Use of transdermal amitriptyline gel in a patient with chronic pain and depression”. Pharmacotherapy 19.2 (1999): 236-239.
- Glinn MA., et al. “Urinary Concentrations of Topically Administered Pain Medications”. Journal of Analytical Toxicology 41.2 (2017): 127-133.
- Kukkar A., et al. “Implications and mechanism of action of gabapentin in neuropathic pain”. Archives of Pharmacal Research 36.3 (2013): 237-251.
- Calandre EP., et al. “Alpha2delta ligands, gabapentin, pregabalin and mirogabalin: a review of their clinical pharmacology and therapeutic use”. Expert Review of Neurotherapeutics 16.11 (2016): 1263-1277.
Citation:
Jan M Keppel Hesselink. “A Rationale for using Gabapentinoids in Topical Analgesic Formulations?” Anaesthesia, Critical Care
and Pain Management 1.1 (2018): 59-62.
Copyright: © 2018 Jan M Keppel Hesselink. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





























 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.