Research Article
Volume 3 Issue 2 - 2018
Applications of Some Natural Active Extracts for Cereal Storage Improvement
1Department of Field Crop Technology Research, Food Technology Research Institute, Agri culture Research Centre, Giza, Egypt
2Department of Food Toxicology and Contaminants, National Research Centre, Dokki, Giza, Egypt
3Arid Lands Cultivation Research Institute, City of Scientific Research and Technological Application, Alexandria, Egypt
2Department of Food Toxicology and Contaminants, National Research Centre, Dokki, Giza, Egypt
3Arid Lands Cultivation Research Institute, City of Scientific Research and Technological Application, Alexandria, Egypt
*Corresponding Author: Abdelazim Sayed Abdelazim, Department of Field Crop Technology Research, Food Technology Research
Institute, Agri culture Research Centre, Giza, Egypt.
Received: July 01, 2018; Published: July 14, 2018
Abstract
Applications of Some Natural Active extracts (Arabic gum, pomegranate peels and Propolis) for Cereal Storage (rice) improvement
were evaluated. Phenolic content was 228.2 ± 17, 87.1 ± 2.3 and 9.7 ± 0.1 mgGAE/g Pomegranate peels extract, Propolis and Arabic
gum, respectively. Total flavonoid ranged from 24.41 ± 1.7 to 42.15 ± 2.83 mg Catechol/g in Propolis and pomegranate peels respectively,
while it was not present in the Arabic gum. Results show the presence of a strong association between the antioxdants activity
against the stable ABTS, DPPH, FRAP radical and total phenol for extracts. Propolis was the most effective extract in either minimal
inhibition concentration (MIC) or minimal bacterial concentrations (MBC) values of propolis were ranged between (1.2–1.5mg/ml
and1.5-1.9mg/ml respectively) which comperad to the standard antibiotic values. Data represented in the order of the antifungal
activities for use substrates as Propolis > pomegranate peels > Arabic gum. The high antibacterial impact of Propolis was recorded on
the bacterial strain of streptococcus aureus. The pomegranate peel extract had the higher reducing effect on Aflatoxin B1 than Propolis
extract and Arabic gum extract (36.39 ± 3.84, 14.93 ± 2.31 and 5.70 ± 4.37(ng/g) respectively, however, for AFG1 the pomegranate
peels recorded higher reducing ratio more than the two other extracts. The coating rice with pomegranate peels extract had the
highest resistance toward the fungal growth during storage than Propolis and Arabic gum extract, respectively. The amount evaluated
of aflatoxins on the rice grains recorded a variation in the extract’s ability to limit the aflatoxins presence on grains. We’re ordering
as pomegranate peels extract > Propolis extract > Arabic gum extract. We can confirm the possibility of using these substances in
functional foods.
Keywords: Arabic gum; Pomegranate peels; Propolis; Natural antioxidant, Antifungal agents; Aflatoxin B1; Aflatoxin G1
Introduction
Mycotoxins, which are fungal secondary metabolites produced during toxigenic fungi metabolism process, are very harmful compound could be found in the feed and food products (Demissie. 2018). Mycotoxins may also the main causes of various types of a health hazard for humans and animals. It consists of a large group of compounds, recently it has reached about four hundred types of toxic materials. Each species of fungi had the ability to excrete one or more of toxic substances, in the same times, one toxin may produce by more than one fungi. It may contaminate food and feed chains which deem the main reason of toxic impacts in the higher creatures when consumed impure commodities (Waliyar., et al. 2008). However, like all over the world, Mycotoxins are a public health worry for Egyptian populations. Cereals, crops, Agricultural products are the major food products suffering from mycotoxin contamination.
Toxigenic fungi are at most out righted by Aspergillus, Penicillium, Alternaria, and Fusarium, however, Aspergillus among the others is very significant fungi, it takes into consideration as a contaminant in either food/feed materials,and it is a plant pathogen. Aspergillus and Penicilliumfungi are mycotoxins producer. Otherwise, toxigenic fungi which recorded to be high grown on cereals are Alternaria, Penicillium,Fusarium, Cladosporium, Stachybotrys, Trichoderma, Acremonium and Paecilomyces (Nooh., et al. 2014; Badr., et al. 2017a). Ultimately, there are four groups of aflatoxins AFB1, AFB2, AFG1, and AFG2 and its two metabolism derivatives AFM1 and AFM2; from which AFB1 is the extreme potency aflatoxins that incentive the health problems to human and animal. Aflatoxins are a toxic compound to human and animal and the main reason for different diseases (Sabry., et al. 2016; Badr., et al. 2017b).
Aflatoxins are particularly a problem in hot and dry cultivated areas, aflatoxins expansion is exacerbated due to the factors affecting of its existence which involves the drought, pests’ infection, and the delayed harvest. As one more point, insufficient drying and poor post-harvest handling are also included as a factors increase the aflatoxin infection (Demissie. 2018). Furthermore, Mycotoxin production by fungi could happen in the storage area causing spoilage in cereals, subsequently actually creating a health problem when it consumed by animals or human. Another hazard factor is the cropping system where previous crop and soil plowing could effect on the resultant crop safety (Groth., et al.1999).
Aflatoxin B1 has classified as group A carcinogens, according to IARC classification report for carcinogenic substances. The modern suggested techniques for aflatoxins reduction are the exclusion of aflatoxin-producing fungi by non-toxigenic strains either simultaneously or with an interval time difference. Different techniques for aflatoxin reducing has applied in the feed using aflatoxin’s binders and additives in animal feed. Also, chemical treatment of contaminated crops is a wonderful method for mycotoxin decreases like the ozone fumigation and treatment with ammonia. Recently, biocontrol of toxigenic fungi to suppress its growth was utilized to reduce the mycotoxin excretion amounts. Moreover, application of probiotic bacteria to reduce aflatoxin uptake record a positive results in many cases. Use of the superabsorbent polymers and the essential oils in mycotoxin binding reported as a successful application, avoiding mycotoxin contamination-hazard in animal feed (Zahra., et al. 2017).
Antioxidants are components which prohibit the oxidation process either in food substances or body fluids. The free radical activity has been involved in cancer, tissue damages, aging, and destruction of DNA. Recently, there has been an increase to examine more substances that can defend against these reactive materials species and play a significant role in disease inhibition (Kapadiya., et al. 2016). Antioxidants play an important role in decreasing mycotoxins impacts in food and feed products (Abdel Razek., et al. 2017; Badr., et al. 2017). Many substances had antioxidation effects such as phenolic components in plant materials, also, flavonoids, plant extracts, and food residual sources. The plant extracts considered a rich material in bio-active substances, it could use to include in the bio-films which applied to reduce mycotoxin contamination that happens in cereal and crops (Shehata., et al. 2017).
The pomegranate (Punicagranatum L.) is a shrub plant mainly grew in the hot and moderate weather. It is widely spread in North Africa, Mediterranean area, India, and Southern East area of Asia. Pomegranate fruits are rich in bioactive substances including the phytochemicals like tannins, anthocyanins, and ellagic acid, and other phenolics. It has been utilized widely in folk medicine in many countries all over the world. Anthocyanin in pomegranate has also is a natural dye which prefered in some types of food products. Moreover, the Propolis is a natural sticky component possessed by the bees from different plants and flowers roots. Chemically the propolis takes notice as a better source of natural antioxidants and antibacterial (Narbona., et al. 2010). Propolis includes an assortment of chemical components like coumarins, steroids, polyphenols, sesquiterpene, quinines, and inorganic materials. Phenolic compounds are the most significant compounds found in propolis, in particular flavonoids and phenolic acids (Falcao., et al. 2010). Propolis has described having various biological effectiveness, including the antibacterial, antifungal, anticancer, antiviral and antitumor characteristics. Consequently, propolis utilized in pharmaceuticals and nutraceuticals to promote health and prevent diseases like pre-cancer, inflammation, hemorrhagic, and chronic diseases (Sforcin., et al. 2011).Arabic Gum is an edible, dried,the sticky exudate substance from the specific types of tree stems and branches. Oral intake of Arabic gum has reported to prolonging various health benefits, it may serve as an antioxidant, anticancer, and antitumor compound (Ali., et al. 2014). Gum Arabic deems an essentially effective due to the powerful antioxidant characteristics.
The aim of the present study was to evaluate the antimicrobial, antifungal, antioxidant, and antimycotoxigenic potency of Pomegranate peels, Propolis, and Arabic gum against some types of food harmful. These hazards included the toxigenic fungi, pathogenic bacteria, and mycotoxins. The result of this study will assist to increase the shelf-life of food and feed products encompass the cereals and its based products. This study is the utilized of food grade extracts with lower cost to enhance the main food products safety and quality. The extraction applied in this study not only safe, cheap, and easy applied; it will also increase the nutritional values of the final products.
Materials and Methods
Materials
Collection of plant materials and sources
Arabic gum (AG) obtained from the Alsamandal for import and export, Batch No.2120, Manufacturing date 10/10/2016, Expired 30/9/2020, From Sudan. Pomegranate peels (PGP) isolated from the Manfaloty Pomegranate fruits purchased from the local market, after collecting it washed, dried, ground and stored in the freezing conditions until applied. Propolis (PP) purchased from the Agricultural Research Center, Beni-Suef Governorate.
Collection of plant materials and sources
Arabic gum (AG) obtained from the Alsamandal for import and export, Batch No.2120, Manufacturing date 10/10/2016, Expired 30/9/2020, From Sudan. Pomegranate peels (PGP) isolated from the Manfaloty Pomegranate fruits purchased from the local market, after collecting it washed, dried, ground and stored in the freezing conditions until applied. Propolis (PP) purchased from the Agricultural Research Center, Beni-Suef Governorate.
Methods
Preparation of extracts
Extracts of pomegranate peels (PGE), propolis (PPE), and Arabic gum (AGE) prepared using isopropyl: water (1:1 v/v) as eco-friendly solvents. Water-isopropyl (v/v) was added to the finely ground powders at a ratio of 1:10 (w/v). The mixture stirred using an overhead stirrer for 50 min at 45°C. The extracts, then separated from the residue by filtration through What man No.4 filter paper. The filtrate concentrated under vacuum at 45°C using a rotary evaporator. The extra solvent was removed by freeze-drying and the dried powder stored at -20°C until further analyses
Preparation of extracts
Extracts of pomegranate peels (PGE), propolis (PPE), and Arabic gum (AGE) prepared using isopropyl: water (1:1 v/v) as eco-friendly solvents. Water-isopropyl (v/v) was added to the finely ground powders at a ratio of 1:10 (w/v). The mixture stirred using an overhead stirrer for 50 min at 45°C. The extracts, then separated from the residue by filtration through What man No.4 filter paper. The filtrate concentrated under vacuum at 45°C using a rotary evaporator. The extra solvent was removed by freeze-drying and the dried powder stored at -20°C until further analyses
Total phenolic content
The Folin–Ciocalteu reagent assay was used to determine the total phenolic content of extracts as described by (Ozsoy., et al. 2008). The total phenolic content expressed in mg Gallic acid equivalents (GAE)/g of sample (mg/g). All determinations performed in triplicate.
The Folin–Ciocalteu reagent assay was used to determine the total phenolic content of extracts as described by (Ozsoy., et al. 2008). The total phenolic content expressed in mg Gallic acid equivalents (GAE)/g of sample (mg/g). All determinations performed in triplicate.
Total flavonoid content
The total flavonoid contents of the pomegranate peels (PGE), propolais (PPE), and Arabic gum (AGE) extracts determined by a colorimetric method described by (Sakanaka., et al. 2005)the absorbance measured at 510 nm. The results were expressed as mg cat./g of sample. All determinations were performed in triplicate.
The total flavonoid contents of the pomegranate peels (PGE), propolais (PPE), and Arabic gum (AGE) extracts determined by a colorimetric method described by (Sakanaka., et al. 2005)the absorbance measured at 510 nm. The results were expressed as mg cat./g of sample. All determinations were performed in triplicate.
DPPH radical scavenging activity
The free radical scavenging activity of pomegranate peels (PGE), propolais (PPE), and Arabic gum (AGE) extracts measured by the DPPH method as proposed by (Turkmen., et al. 2005). The measured conditions recorded at 517 nm with a spectrophotometer.
The free radical scavenging activity of pomegranate peels (PGE), propolais (PPE), and Arabic gum (AGE) extracts measured by the DPPH method as proposed by (Turkmen., et al. 2005). The measured conditions recorded at 517 nm with a spectrophotometer.
ABTS cat ion decolorization assay.
ABTS radical assay used to evaluate the ability to scavenge free ABTS radicals, based on the protocol of (Re., et al. 1999). Absorbance readings measured using a spectrophotometer at 734 nm. Results were expressed as μ mol of trolox equivalents (TE) per g sample from a standard curve developed with Trolox. All determinations performed in triplicate.
ABTS radical assay used to evaluate the ability to scavenge free ABTS radicals, based on the protocol of (Re., et al. 1999). Absorbance readings measured using a spectrophotometer at 734 nm. Results were expressed as μ mol of trolox equivalents (TE) per g sample from a standard curve developed with Trolox. All determinations performed in triplicate.
Ferric Reducing Ability (FRAP) assay
The FRAP assay was done according to according to (Hwang., et al. 2014). Readings of the colored product, absorbent measured at 593 nm. The standard curve was prepared using Trolox and the results were expressed as mMTorlox equivalent (TE/g sample).
The FRAP assay was done according to according to (Hwang., et al. 2014). Readings of the colored product, absorbent measured at 593 nm. The standard curve was prepared using Trolox and the results were expressed as mMTorlox equivalent (TE/g sample).
Determination of minimal inhibition concentration of extracts.
The minimal inhibitory concentration (MIC) value is determined as the lowest concentration of extract that didn't give any visible bacterial growth. Each assay carried out in triplicate. The MIC test quantified the antimicrobial activity of the extract, it was done as the method described by (Sakanaka., et al. 2005).
The minimal inhibitory concentration (MIC) value is determined as the lowest concentration of extract that didn't give any visible bacterial growth. Each assay carried out in triplicate. The MIC test quantified the antimicrobial activity of the extract, it was done as the method described by (Sakanaka., et al. 2005).
Determination of minimal fungicidal concentration of extracts
The hypha growth inhibition test can be used to determine the antifungal activity of the extracts against fungal strains as previously described by (Picman., et al. 1990).The concentration required to give 50% inhibition of hyphen growth IC50will be calculated from the regression equation. Nystatin can be used as a positive control.
The hypha growth inhibition test can be used to determine the antifungal activity of the extracts against fungal strains as previously described by (Picman., et al. 1990).The concentration required to give 50% inhibition of hyphen growth IC50will be calculated from the regression equation. Nystatin can be used as a positive control.
Determination of several types of extraction on fungal growth.
Yeast extract sucrose (YES) used to examine the effect of each extraction method which may include several components of the fungal growth of Aspergillus parasiticus ATTC 15517, also the powder of the three materials examined by using 0.5g of each as additional material in liquid media. The liquid media of YES consists of 15g yeast extract, 150g sugar, and completely to one literusing de-ionize double distilled water. The inhibition effect represented as the loss of the fungal growth in the presence of extracts against the control growth.
Yeast extract sucrose (YES) used to examine the effect of each extraction method which may include several components of the fungal growth of Aspergillus parasiticus ATTC 15517, also the powder of the three materials examined by using 0.5g of each as additional material in liquid media. The liquid media of YES consists of 15g yeast extract, 150g sugar, and completely to one literusing de-ionize double distilled water. The inhibition effect represented as the loss of the fungal growth in the presence of extracts against the control growth.
Preparation of standards for aflatoxin
The standard of Aflatoxins received as dry films or crystals to container of dry aflatoxins B, G, add a volume of the following solvents: methanol: acetonitrile (9:1) calculated to give a concentration as ng/ml.
The standard of Aflatoxins received as dry films or crystals to container of dry aflatoxins B, G, add a volume of the following solvents: methanol: acetonitrile (9:1) calculated to give a concentration as ng/ml.
HPLC Chromatography
One hundred microliters of the samples were injected into the HPLC column. The mobile phase was an acetonitrile: water: acetic acid solution (51:47:2, v/v). The flow rate was 1 ml/min. High performance liquid chromatography (HPLC) system consisted of Waters Binary Pump Model1525, Model Waters 1500 Rheodyne Manual Injector, Waters 2475 Multi-Wavelength Fluorescence Detector, and a data workstation with software Breeze. A Phenomenex ColumnC18, dimensions: 250 × 4.6 mm, particle size: 5 µm, from Waters Corporation (USA) as well as Microfiber Filters, 11 cm, product ID: 31955, VICAM Company (Sweden), were used.
One hundred microliters of the samples were injected into the HPLC column. The mobile phase was an acetonitrile: water: acetic acid solution (51:47:2, v/v). The flow rate was 1 ml/min. High performance liquid chromatography (HPLC) system consisted of Waters Binary Pump Model1525, Model Waters 1500 Rheodyne Manual Injector, Waters 2475 Multi-Wavelength Fluorescence Detector, and a data workstation with software Breeze. A Phenomenex ColumnC18, dimensions: 250 × 4.6 mm, particle size: 5 µm, from Waters Corporation (USA) as well as Microfiber Filters, 11 cm, product ID: 31955, VICAM Company (Sweden), were used.
Detoxification methods.
The extracts prepared by using aqueous isopropyl, the extracted materials concentrated to near dried, then freeze-dried used to convert it as powder materials, 5mg of each extract concentrate was dissolved in 0.5 mL in Di-methyl sulphoxide (DMSO), this value used to inject in the plate wells or in the plate disks as a detoxification application against either pathogenic bacteria or toxigenic fungi (Esper., et al. 2014, Balouiri., et al. 2016).
The extracts prepared by using aqueous isopropyl, the extracted materials concentrated to near dried, then freeze-dried used to convert it as powder materials, 5mg of each extract concentrate was dissolved in 0.5 mL in Di-methyl sulphoxide (DMSO), this value used to inject in the plate wells or in the plate disks as a detoxification application against either pathogenic bacteria or toxigenic fungi (Esper., et al. 2014, Balouiri., et al. 2016).
Total fungal count of toxigenic fungi inoculated to rice with several treatments before and after 60 days of storage.
To apply the results of the study, the antifungal ability of each extract of PGE, PPE, and AGE were applied on rice grain samples. A rice samples equal to 150g were brushed with a water solution of each extract for 3 min. After that, the samples were dried in hot air oven at 45°C for 5-6h to reach 11% moisture content. The samples of the three extracts treatments, side of the control without treatment were packed in a sterilized polyethylene bag. Each type of extract treatment was inoculated by nine strains of mycotoxigenic fungi (one strain/one bag/one treatment). The initial count of each inoculated strain was recorded as before treatment and at the end of the storage period (60 days) the final count of each strain recorded as after treatment value.
To apply the results of the study, the antifungal ability of each extract of PGE, PPE, and AGE were applied on rice grain samples. A rice samples equal to 150g were brushed with a water solution of each extract for 3 min. After that, the samples were dried in hot air oven at 45°C for 5-6h to reach 11% moisture content. The samples of the three extracts treatments, side of the control without treatment were packed in a sterilized polyethylene bag. Each type of extract treatment was inoculated by nine strains of mycotoxigenic fungi (one strain/one bag/one treatment). The initial count of each inoculated strain was recorded as before treatment and at the end of the storage period (60 days) the final count of each strain recorded as after treatment value.
Application of some natural active extracts on rice grains as cereal food preservatives
To apply the results of the study, the antifungal ability of each extract of PGE, PPE, and AGE were applied on rice grain samples. A rice samples equal to 150g were brushed with a water solution of each extract for 3 min. After that, the samples were dried in hot air oven at 45°C for 5-6h to reach 11% moisture content. The samples of the three extracts treatments, side of the control without treatment were packed in a sterilized polyethylene bag. The bags of the three treatments and the control were inoculated with a high producing toxic strain of Aspergillus parasiticus ITEM 11 and incubated at 25°C for 15 days. The result of aflatoxins amount that excreted on the rice seeds were evaluated using HPLC techniques.
To apply the results of the study, the antifungal ability of each extract of PGE, PPE, and AGE were applied on rice grain samples. A rice samples equal to 150g were brushed with a water solution of each extract for 3 min. After that, the samples were dried in hot air oven at 45°C for 5-6h to reach 11% moisture content. The samples of the three extracts treatments, side of the control without treatment were packed in a sterilized polyethylene bag. The bags of the three treatments and the control were inoculated with a high producing toxic strain of Aspergillus parasiticus ITEM 11 and incubated at 25°C for 15 days. The result of aflatoxins amount that excreted on the rice seeds were evaluated using HPLC techniques.
Statistical evaluation.
The obtained results were evaluated statistically using analysis of variance (One way ANOVA analysis) using SPSS 16.0 as reported by Mc Clave and Benson (McClave., et al. 1991).
The obtained results were evaluated statistically using analysis of variance (One way ANOVA analysis) using SPSS 16.0 as reported by Mc Clave and Benson (McClave., et al. 1991).
Results and Discussions
Total phenolic and flavonoid content
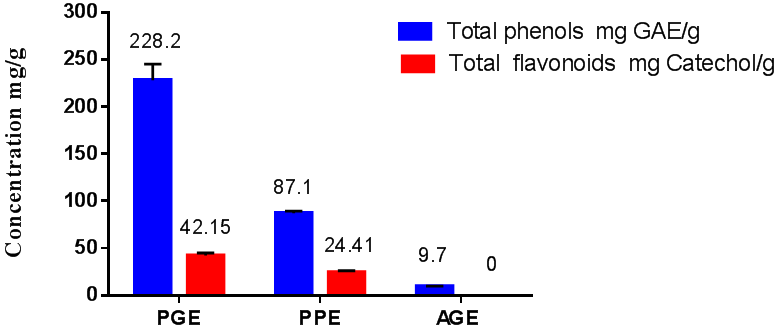
The total phenolic content (TPC) and total flavonoid contents (TFC) of the three extracts comprised PGE, PPE, and AGE have evaluated. The results were recorded as shown inFigure 1. The TPH was 228.2 ± 17, 87.1 ± 2.3, 9.7 ± 0.1 mg GAE/g for PGE, PPE, and AGE, respectively. However, The TFC recorded at values of 24.41 ± 1.7 and 42.15 ± 2.83 mg Catechol/g for Propolis and pomegranate peels, respectively; otherwise, it was not detected in the Arabic gum. Health benefits of the bioactive molecules included the flavonoids was exhibited and its potent antioxidant impact had proved (Orak ., et al. 2012). The TPH and the TF were significantly different among different extracts as reported in Figure 1. Our results are compatible with several recent studies (Orak., et al. 2012; Mohdaly., et al. 2015 and Ramadan., et al. 2016), these studies reported the highest values of TPH in pomegranate peels and Propolis. There are various reports of TPH and TFC in pomegranate peels with a wide difference in the published results is remarked. This could be the result of geographical, cultivar (Lu., et al. 2008; Fawole., et al. 2012; Hasnaoui., et al. 2014). Variations in the results might be also due to the different extraction procedure used in the experiments, particularly due to the type and the concentration of the starting extraction solvent. (Fawole., et al. 2012) noted that the aqueous ethanol was effective in the TPH extraction of PGP with values ranged from 180 to 290 mg GAE/g. The TPH in PP ranged from 79.8 to 156.3 μg/g, the average being 119.5 μg/g.As a result of this discussion, aqueous solvent extraction technique could be a superior system for the effective bioactive compound extraction from PGP.
The total phenolic content (TPC) and total flavonoid contents (TFC) of the three extracts comprised PGE, PPE, and AGE have evaluated. The results were recorded as shown inFigure 1. The TPH was 228.2 ± 17, 87.1 ± 2.3, 9.7 ± 0.1 mg GAE/g for PGE, PPE, and AGE, respectively. However, The TFC recorded at values of 24.41 ± 1.7 and 42.15 ± 2.83 mg Catechol/g for Propolis and pomegranate peels, respectively; otherwise, it was not detected in the Arabic gum. Health benefits of the bioactive molecules included the flavonoids was exhibited and its potent antioxidant impact had proved (Orak ., et al. 2012). The TPH and the TF were significantly different among different extracts as reported in Figure 1. Our results are compatible with several recent studies (Orak., et al. 2012; Mohdaly., et al. 2015 and Ramadan., et al. 2016), these studies reported the highest values of TPH in pomegranate peels and Propolis. There are various reports of TPH and TFC in pomegranate peels with a wide difference in the published results is remarked. This could be the result of geographical, cultivar (Lu., et al. 2008; Fawole., et al. 2012; Hasnaoui., et al. 2014). Variations in the results might be also due to the different extraction procedure used in the experiments, particularly due to the type and the concentration of the starting extraction solvent. (Fawole., et al. 2012) noted that the aqueous ethanol was effective in the TPH extraction of PGP with values ranged from 180 to 290 mg GAE/g. The TPH in PP ranged from 79.8 to 156.3 μg/g, the average being 119.5 μg/g.As a result of this discussion, aqueous solvent extraction technique could be a superior system for the effective bioactive compound extraction from PGP.
Figure 1: Total phenolic and total flavonoid content for
extracts of pomegranate peels,Propolis and Arabic gum.
The antioxidant activity of the PGE, PPE, and AGE extracts using DPPH, ABTS, and FRAP assays
Antioxidant activity of three extracts of PGE, PPE, and AGE measured using three parallel test assays, the results are presented in Table 1. The values indicate that; the IC50 of extracts is ordering descending at P = 0.05 from PGE to AGE. The antioxidant potency of PGE, PPE, and AGE evaluated using three different assays, these were DPPH, ABTS, and FRAP reducing ability. Among the extracts analyzed, PGE recorded the greatest antioxidant activity by the three methods. Low ICIC50 corresponds to a strong inhibitory capacity of DPPH, ABTS, FRAP radical, the highest IC50 is recorded for the AG.
Antioxidant activity of three extracts of PGE, PPE, and AGE measured using three parallel test assays, the results are presented in Table 1. The values indicate that; the IC50 of extracts is ordering descending at P = 0.05 from PGE to AGE. The antioxidant potency of PGE, PPE, and AGE evaluated using three different assays, these were DPPH, ABTS, and FRAP reducing ability. Among the extracts analyzed, PGE recorded the greatest antioxidant activity by the three methods. Low ICIC50 corresponds to a strong inhibitory capacity of DPPH, ABTS, FRAP radical, the highest IC50 is recorded for the AG.
In a previous study; the PGE had the highest antioxidant activity among the peels, pulp and seed fractions of 28 kinds of fruits commonly consumed in China, as determined by FRAP assay that described in the study of (Guo., et al. 2003). (While Pagliarulo., et al. 2016) demonstrated that the methanol extract of pomegranate peels had the highest antioxidant activity among ethyl acetate, methanol, and water extracts. Moreover, (Li., et al. 2006) reported the aqueous methanol to be more effective than each solvent alone. The antioxidant activity potential of Propolis may be due to the presence of diverse phytochemical compounds, mainly flavonoids and phenolic acids. (Mohdaly., et al. 2015). The decrease of DPPH radical by the different plant extracts has been attributed by many authors, at the presence of phenolic compounds which yield simply to reduce protons (Li., et al. 2006). Our results recorded the presence of a strong association between the scavenging activities against the stable ABTS, DPPH, FRAP radical and total phenol for extracts. This could the main reason in the antimicrobial, antifungal and antimycotoxigenic impact of these types of extracts. These results are in agreement with results of (Abdelazim. 2017) who found that Silymarin can be used as natural antioxidants and antimicrobial activity in the food industry.
| Assays | IC50 values (µg/ml) | ||
| Pomegranate peels | Propolis | Arabic gum | |
| DPPH | 1.47 ± 0.4 | 3.28 ± 0.74 | 6.1 ± 1.3 |
| ABTS | 1.65 ± 0.32 | 2.86 ± 1.2 | 5.28 ± 0.97 |
| FRAP | 0.81 ± 0.34 | 2.91 ± 0.67 | 5.92 ± 0.84 |
Table 1: Comparative evaluation of Pomegranate peels, Arabic gum and Propolis for antioxidant activity using DPPH, ABTS and FRAP radical scavenging
IC50: Concentration of sample required for 50% inhibition. Results are expressed as mean of three experiments ± SD
IC50: Concentration of sample required for 50% inhibition. Results are expressed as mean of three experiments ± SD
Minimal inhibitory and minimal bacterial concentration
The MIC values of the three extracts for the raw materials of PGPE, PPE, and AGE evaluated to explain the antimicrobial impact. Moreover, minimal bacterial concentration (MBC) were determined to explore the ability of these extracts to apply as antibacterial components in food productions. Four pathogenesis bacterial strains were applied to examine the inhibition of its growth in the presence of these GRAS extracts, Amoxicillin antibiotic was utilized as a standard reference to compare the antibacterial effects. Represented as the data in Table 2, Propolis was the most effective extract in either minimal inhibition concentration. MIC and MBC values of propolis were ranged between (1.2–1.5mg/ml and1.5-1.9mg/ml respectively) which considered closed to the standard antibiotic values. Among the three extracts, Arabic gum extraction showed the lowest values either for MIC or the MBC on the pathogenic bacterial strains in this experiment. The results of MIC and MBC values of the three types of the extracts were applied against three types of pathogenic strains, these strains were Pseudomonase aerugnosa ATCC 9027, Enterococcus faecalies ATCC47077, and Staphylococcus aureus NCTC 10788. The strain of Enterococcus faecalies ATCC47077 recorded a high resistance value either in MIC or in MBC for the three types of extracts.
The MIC values of the three extracts for the raw materials of PGPE, PPE, and AGE evaluated to explain the antimicrobial impact. Moreover, minimal bacterial concentration (MBC) were determined to explore the ability of these extracts to apply as antibacterial components in food productions. Four pathogenesis bacterial strains were applied to examine the inhibition of its growth in the presence of these GRAS extracts, Amoxicillin antibiotic was utilized as a standard reference to compare the antibacterial effects. Represented as the data in Table 2, Propolis was the most effective extract in either minimal inhibition concentration. MIC and MBC values of propolis were ranged between (1.2–1.5mg/ml and1.5-1.9mg/ml respectively) which considered closed to the standard antibiotic values. Among the three extracts, Arabic gum extraction showed the lowest values either for MIC or the MBC on the pathogenic bacterial strains in this experiment. The results of MIC and MBC values of the three types of the extracts were applied against three types of pathogenic strains, these strains were Pseudomonase aerugnosa ATCC 9027, Enterococcus faecalies ATCC47077, and Staphylococcus aureus NCTC 10788. The strain of Enterococcus faecalies ATCC47077 recorded a high resistance value either in MIC or in MBC for the three types of extracts.
On the other hands, some of the previous studies reported an antibacterial activity of ethanolic extracts of the Propolis, the results manifested a high antibacterial action against gram-positive bacteria with a MIC ranging from 0.2 to 0.78 mg/mL, also it was reported an elevated antibacterial activity against gram-negative bacteria Salmonellaenterica (Mohdaly., et al. 2015 and Ramadan., et al. 2016)
| Strains | Pomegranate peels | Propolis | Arabic gum | Amoxicillin | |
| Concentration (mg/mL) | |||||
| Pseudomonase aerugnosa ATCC 9027 | MIC* | 3.2 | 1.4 | 7.4 | 0.1 |
| Pseudomonase aerugnosa ATCC 9027 | MBC** | 3.6 | 1.7 | 8.3 | 0.2 |
| Enterococcus faecalies ATCC 47077 | MIC | 3.7 | 1.5 | 7.5 | 0.1 |
| Enterococcus faecalies ATCC 47077 | MBC | 3.9 | 1.9 | 8.9 | 0.1 |
| Staphylococcus aureus NCTC 10788 | MIC | 3.4 | 1.2 | 7.2 | 0.1 |
| Staphylococcus aureus NCTC 10788 | MBC | 3.7 | 1.5 | 8.3 | 0.2 |
Table 2: Minimum inhibitory concentration for Pomegranate peels, Arabic gum and Propolis extracts.
*MIC minimal inhibitory concentration **MBCminimal bacterial concentration
*MIC minimal inhibitory concentration **MBCminimal bacterial concentration
Antifungal effect of different extraction types on Aspergillus parasiticus
Antifungal effect of several types of the extraction methods of Arabic gum, Propolis, and pomegranate peelswere applied on YES media against the fungal growth of Aspergillus parasiticus ATTC 15517 showed in Table 3. The extracts were ass to the YES liquid media with a concentration of 10mg/ml for each material, also the powder of the three materials was examined by using 0.5g of each as additional material in liquid media. The results represented a good inhibition impact of the pomegranate peels on the fungal growth for the aqueous extraction methods as well as for the powder addition in the media
Antifungal effect of several types of the extraction methods of Arabic gum, Propolis, and pomegranate peelswere applied on YES media against the fungal growth of Aspergillus parasiticus ATTC 15517 showed in Table 3. The extracts were ass to the YES liquid media with a concentration of 10mg/ml for each material, also the powder of the three materials was examined by using 0.5g of each as additional material in liquid media. The results represented a good inhibition impact of the pomegranate peels on the fungal growth for the aqueous extraction methods as well as for the powder addition in the media
| Extracts (10mg/ml) | Control | Arabic gum | Propolis | Pom Peels |
| Water | 4.58 ± 1.47* | 4.76 ± 0.49 | 3.47 ± 0.54 | 3.41 ± 2.56 |
| Isopropanol:waterer (1:1) | 4.58 ± 1.47 | 5.11 ± 0.71 | 3.33 ± 0.51 | 3.23 ± 0.63 |
| Hexane | 4.58 ± 1.47 | 4.31 ± 1.65 | 3.92 ± 0.72 | 4.91 ± 0.44 |
| Petroleum | 4.58 ±1.47 | 4.43 ± 0.35 | 3.72 ± 0.27 | 4.83 ± 0.36 |
| powder 0.5g/L | 4.58 ± 1.47 | 4.83 ± 0.87 | 1.78 ± 0.73 | 2.37 ± 0.66 |
Table 3: Effect of powder Pomegranate peels, Propolis and it’s their extracts on fungus growth (g).
*Data were represented as mean of values ± SD. The weight of fungal growth on liquid media is recorded in grams.
*Data were represented as mean of values ± SD. The weight of fungal growth on liquid media is recorded in grams.
Antifungal activity of pomegranate peels, propolis extracts and Arabic gum against tested fungus strains (mm showed in Table 4. In order to the need to ensure the results, the antifungal of these plant materials, extracts were tested by other antifungal method. Plate inhibition zone assay of the extracts against four toxigenic fungi strains were applied to explore the variations recorded between the several materials in the antifungal effect, this will be useful to determine the suitable amounts which aimed to be applied as food additive to enhance the good qualities of food against the fungal contaminations. The data represented in the order of the antifungal activities for used substrates as Propolis > Pomegranate peels > Arabic gum. The extract of Propolis showed a closed result to the standard antifungal compound (Nystatine). The high antifungal impact of Propolis was recorded on the Fusarium fungi.
| Materials | Aspergillus paracitcus ATCC 15517 | Penicillium citrinum NRRL 1841T | Alternaria alternata ATTC 96151 |
Fusarium graminearum ITEM 124 |
| Diameters of Inhibition Zones (mm) | ||||
| Pomegranatepeels (10mg/ml) | 11 ± 1.2* | 14 ± 1.5 | 13 ± 0.6 | 11 ± 1 |
| Propolis (10mg/ml) | 22 ± 1.4 | 24 ± 1.2 | 23 ± 2.1 | 27 ± 1.2 |
| Arabic gum (0.5g/ L) | 7 ± 1.5 | 3 ± 1 | 1 ± 0.6 | 5 ± 0.7 |
| Nystatine (0.15ul/ml) | 35 ± 1.1 | 35 ± 1.3 | 35 ± 1.1 | 35 ± 1.2 |
Table 4: Antifungal activity of pomegranate peels, Propolis extracts and Arabic gum against tested fungus strains (mm).
*Data were represented as mean of values ± SD. Each value represents the mean of three replicates
*Data were represented as mean of values ± SD. Each value represents the mean of three replicates
Antibacterial activity of the extracts of pomegranate peels, Propolis and Arabic gum extracts.
Antibacterial characteristics of the three materials, extracts were measured as an inhibition zone on them against the bacterial growth of four pathogenic strains on potato dextrose agar supplemented by Nystatine as antifungal antibiotic to suppress the fungi and the results showed in Table 5. Plate inhibition zone assay of the extracts against four bacterial strains were applied to explore the variations recorded between the several materials in the antibacterial impact, this will help to evaluate the application of these extracts during the food manufacturing to support the good qualities of food products as well as its expected application in food commodities coating against the bacterial spoilage. The data represented in the order of antibacterial activities for use substrates as Propolis > pomegranate peels > Arabic gum. The extract of Propolis showed a closed result to the standard antifungal compound (Amoxicillin). The high antibacterial impact of Propolis was recorded on the bacterial strain of Streptococcus aureus.
Antibacterial characteristics of the three materials, extracts were measured as an inhibition zone on them against the bacterial growth of four pathogenic strains on potato dextrose agar supplemented by Nystatine as antifungal antibiotic to suppress the fungi and the results showed in Table 5. Plate inhibition zone assay of the extracts against four bacterial strains were applied to explore the variations recorded between the several materials in the antibacterial impact, this will help to evaluate the application of these extracts during the food manufacturing to support the good qualities of food products as well as its expected application in food commodities coating against the bacterial spoilage. The data represented in the order of antibacterial activities for use substrates as Propolis > pomegranate peels > Arabic gum. The extract of Propolis showed a closed result to the standard antifungal compound (Amoxicillin). The high antibacterial impact of Propolis was recorded on the bacterial strain of Streptococcus aureus.
| Materials | Streptococcus aureus NCTC 10788 |
Bacillus cereus ATCC 11778 |
Salmonella typhi ATCC 14028 |
E. coli ATCC11229 |
| Diameters of Inhibition Zones (mm) | ||||
| Pomegranate peels(10mg/ml) | 15 ± 0.3 | 16 ± 0.6 | 13 ± 0.6 | 15 ± 0.7 |
| Propolis (10mg/ml) | 24 ± 1.1 | 23 ± 0.6 | 20 ± 0.34 | 21 ± 0.66 |
| Arabic gum (0.5g/ L) | 12 ± 1.7 | 10 ± 0.6 | 8 ± 1.2 | 8 ± 0.71 |
| Amoxicyllin (0.2ul/ml) | 32 ± 0.34 | 31 ± 0.61 | 29 ± 1.2 | 29 ± 0.56 |
Table 5: Antibacterial activity of pomegranate peels, Propolis and Arabic gum extracts against tested bacterial strains (mm).
Data were represented as mean of values ± SD. Each value represents the mean of three replicates. In a previous study, The PP reported to had an antibacterial activity against some strains of testing pathogens such as; Salmonella enteritidis ATCC 13076, Staphylococcus aureus ATCC 13565 Escherichia coli ATTC 25923, Listeria monocytogenes and Bacillus subtilis NCTC 8236 (Attalla., et al. 2007). Antimicrobial activity of PP also reported by using several types of solvents (Ethanol, Acetone, Toluene). Otherwise, the change in the pH values of the resultant extract at three different pH values (6, 7, 8) showed a widely range of PP antibacterial activity (Salman. 2016). This result agrees with (Chacko., et al. 2014); (Labyad., et al. 2016) they described exhibited antibacterial activity of PGP against various Gram-positive and Gram-negative bacteria of pomegranate peels was characterized by the highest antibacterial activity against of Shigella strain of pathogenic bacterial (Vodnar., et al. 2017).
Notwithstanding; (Rather., et al. 2010) extracted phenolics from dried powdered PP with various solvents (methanol, ethyl acetate, chloroform, and water) and revealed the antimicrobial activity of methanolic extract against Escherichia coli, Pseudomonas sp., Klebsiella sp., vancomycin and methicillin resistant Staphylococcus aureus.
Anti-aflatoxigenic effect of Pomegranate peels, Propolis and Arabic gum extracts against aflatoxins in liquid media
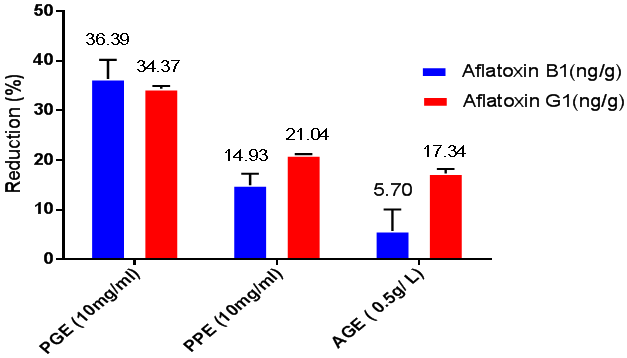
Reducing the impact of iso-propanol-water extracts of Pomegranate peels, Propolis and Arabic gum on aflatoxin B1 and aflatoxin G1 were exanimated in the YES fungal growth of liquid media, the amount of AFB1 and AFG1 were determined in the control flask of Aspergillus paraciticus strain ATCC 11517 and the results illustrated in Figure 2.The toxin amount was indicated the control aflatoxins amount of AFB1 and AFG1. While, other flasks were supported by the extracted materials at a concentration of 10mg/ml in the growth media. Using the apparatus of HPLC, the reducing amount of aflatoxins was recorded against the amount that excreted in the control flask. The reducing amount here was reflected either the inhibition effect of the extracted material on the fungal excretion metabolites or it could be referred to the chemical reaction happen during the extraction present in growth media. The PGE had the higher reducing effect on Aflatoxin B1 and G1 more than PPE and AGE (36.39 ± 3.84, 14.93 ± 2.31, 5.70 ± 4.37(ng/g) and (34.37 ± 0.6 than 21.04 ± 0.18 and 17.34 ± 0.88 (ng/g)) respectively, however, for AFG1 the recorded the higher reducing ratio for each of the extracts compared to control. The AGE recorded as the lower effect than PGEand PPE) 52.9 ± 0.88than 42.56 ± 0.6 and 50.53 ± 0.18 (ng/g (respectively on both Aflatoxin B1and Aflatoxin G1. Notwithstanding, the recovery for the aflatoxins from AG powder precipitated in the YES media after the experimental time was not applied to explain if the toxin could recover from the powder or not. Maybe the effect of the pomegranate peel extract is higher than the other extracts to cause the high content of polyphenolic compound.
Reducing the impact of iso-propanol-water extracts of Pomegranate peels, Propolis and Arabic gum on aflatoxin B1 and aflatoxin G1 were exanimated in the YES fungal growth of liquid media, the amount of AFB1 and AFG1 were determined in the control flask of Aspergillus paraciticus strain ATCC 11517 and the results illustrated in Figure 2.The toxin amount was indicated the control aflatoxins amount of AFB1 and AFG1. While, other flasks were supported by the extracted materials at a concentration of 10mg/ml in the growth media. Using the apparatus of HPLC, the reducing amount of aflatoxins was recorded against the amount that excreted in the control flask. The reducing amount here was reflected either the inhibition effect of the extracted material on the fungal excretion metabolites or it could be referred to the chemical reaction happen during the extraction present in growth media. The PGE had the higher reducing effect on Aflatoxin B1 and G1 more than PPE and AGE (36.39 ± 3.84, 14.93 ± 2.31, 5.70 ± 4.37(ng/g) and (34.37 ± 0.6 than 21.04 ± 0.18 and 17.34 ± 0.88 (ng/g)) respectively, however, for AFG1 the recorded the higher reducing ratio for each of the extracts compared to control. The AGE recorded as the lower effect than PGEand PPE) 52.9 ± 0.88than 42.56 ± 0.6 and 50.53 ± 0.18 (ng/g (respectively on both Aflatoxin B1and Aflatoxin G1. Notwithstanding, the recovery for the aflatoxins from AG powder precipitated in the YES media after the experimental time was not applied to explain if the toxin could recover from the powder or not. Maybe the effect of the pomegranate peel extract is higher than the other extracts to cause the high content of polyphenolic compound.
Effect of natural extracts on the growth of fungi during storage period of rice.
To gain the benefits of using these extracts as antimicrobial and antifungal components, it has applied as a brushing coat on the rice grains then the grains dried before packed in polyethylene bags. These bags stored for 60 days before the fungal count calculated. At the initial time; the grains inoculated by one strain of fungi for each bag and the log CFU/g of rice recorded as (before value), while at the end of the storing period the log CFU/g of rice evaluated as (after value). The reducing values of the log CFU/g indicated the power of the extract type as anti-mycotic material which help to enhancement the grain safety during the storage period. The results in Table 6 and Figure (3) showed a high powerful of each extract to reduce the log CFU/g of fungi during the storage period. The PGE coating material appeared as an excellent coated material against the nine strains of toxigenic fungi. while AGE exhibited the lowest type of coated material to resist the fungal growth on the stored rice. These results support the application of PGE as a cheap source of bioactive substances to reduce the fungal infection in the stored grain when it is applying as coating film. The results gained from this experiment was in agreement with (Shehata., et al. 2017)
To gain the benefits of using these extracts as antimicrobial and antifungal components, it has applied as a brushing coat on the rice grains then the grains dried before packed in polyethylene bags. These bags stored for 60 days before the fungal count calculated. At the initial time; the grains inoculated by one strain of fungi for each bag and the log CFU/g of rice recorded as (before value), while at the end of the storing period the log CFU/g of rice evaluated as (after value). The reducing values of the log CFU/g indicated the power of the extract type as anti-mycotic material which help to enhancement the grain safety during the storage period. The results in Table 6 and Figure (3) showed a high powerful of each extract to reduce the log CFU/g of fungi during the storage period. The PGE coating material appeared as an excellent coated material against the nine strains of toxigenic fungi. while AGE exhibited the lowest type of coated material to resist the fungal growth on the stored rice. These results support the application of PGE as a cheap source of bioactive substances to reduce the fungal infection in the stored grain when it is applying as coating film. The results gained from this experiment was in agreement with (Shehata., et al. 2017)
| Strain of Fungi | Control | Arabic Gum | Propolis | Pomegranate Peels | |
| Before | After | After 60 Days of Storage | |||
| Aspergillus niger ITEM 3856 | 2.322 | 6.204 | 5.50 | 4.77 | 3.92 |
| Aspergillus parasiticus ITEM 11 | 2.23 | 6.34 | 6.11 | 4.92 | 3.77 |
| Aspergillus carbonarius ITEM5010 | 2.36 | 6.079 | 5.61 | 4.55 | 3.86 |
| Aspergillus flavus ITEM 698 | 2.20 | 5.76 | 5.32 | 3.74 | 2.81 |
| Aspergillus oryzae ITEMB5 | 2.11 | 5.63 | 4.78 | 4.59 | 2.83 |
| Fusarium oxysporum ITEM 12591 | 2.20 | 4.86 | 4.44 | 3.61 | 2.93 |
| Aspergillus niger ITEM 3856 | 2.27 | 5.95 | 1.633 | 1.43 | 1.73 |
| Fusarium solani ITEM 250 | 2.04 | 4.71 | 1.91 | 1.53 | 1.83 |
| Penicillium chrysogenum ATCC 10106 | 2.14 | 5.83 | 1.96 | 1.79 | 2.70 |
Table 6: Effect of natural extracts on the growth of fungi during storage period (60 days.) of rice.
Value represented at the mean value of five replecates. The LSD = 2.189
Value represented at the mean value of five replecates. The LSD = 2.189
Figure 3: Effect of natural extracts on the growth of against A. parasiticus ITEM 11
after 15 days of rice. (1) Control; (2) Arabic gum; (3) proplies; (4) pomegranate peels.
Effect of natural extracts on the reduce Aflatoxins during storage period of rice
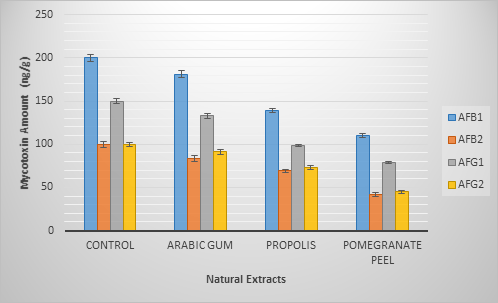
As an application to convert the results of this experiment to increase the agriculture, sustainable production and increase the food safety, the extracts of PGE, PPE, and AGE had applied to coat the rice grains and inoculated by A. parasiticus ITEM 11 strain which recorded as a high producing strain of the four types of aflatoxins. According to the recorded results; PGE was the effective extract that able to reduce the toxin excretion on the stored rice grain during the optimum time of aflatoxin excretion by the fungi (about 15 days) Figure 4. The amount evaluated of aflatoxins on the rice grains recorded a variation in the extract,s ability to limit the aflatoxins presence on grains. The reducing ratio were ordering as PGE, PPE, AGE, respectively.
As an application to convert the results of this experiment to increase the agriculture, sustainable production and increase the food safety, the extracts of PGE, PPE, and AGE had applied to coat the rice grains and inoculated by A. parasiticus ITEM 11 strain which recorded as a high producing strain of the four types of aflatoxins. According to the recorded results; PGE was the effective extract that able to reduce the toxin excretion on the stored rice grain during the optimum time of aflatoxin excretion by the fungi (about 15 days) Figure 4. The amount evaluated of aflatoxins on the rice grains recorded a variation in the extract,s ability to limit the aflatoxins presence on grains. The reducing ratio were ordering as PGE, PPE, AGE, respectively.
Figure 4: Aflatoxins Amounts on different rice treated samples inoculated by
A. parasiticus ITEM 11 strain after 15 days of storage.
A. parasiticus ITEM 11 strain after 15 days of storage.
Conclusion
Pomegranate peels, Propolis and Arabic gum had considerable levels of polyphenolic compounds. Their chemical characteristics induce in general a strong bactericidal action against Pseudomonase aerugnosa ATCC 9027 and Staphylococcus aureus NCTC 10788, Propolis, Pomegranate peels and Arabic gum had a reducing effect on Aflatoxin B1 and Aflatoxin G1 (ng/g). Presented results of this study demonstrated high biological potential of these extracts. Statistical analysis showed high correlation among the contents of investigating classes of compounds and biological activity of prepared extracts. All in all, these results confirmed that extract of Pomegranate peels, Propolis and Arabic gum might indeed be a potential source of biological compounds. We can could be reported can use the Propolis, Pomegranate peels and Arabic gum as plant natural antioxidant, antimicrobial agents and their toxics and as functional foods.
References
- Abdelazim SAA. “Effect of Silymarin as Natural Antioxidants and Antimicrobial Activity”. Egyptian Journal of Agricultural Research 95.2 (2017): 725-737.
- Abdel-Razek A., et al. “Characterization of Olive Oil By-products: Antioxidant Activity, Its Ability to Reduce Aflatoxigenic Fungi Hazard and Its Aflatoxins”. Australian Road Research Board 14.5 (2017): 1-14.
- Abuarra A., et al. “Fabrication and characterization of gum Arabic bonded Rhizophora spp”. Particleboards Materials and Design60 (2014): 108-115.
- Ali A., et al. “Composite effect of Propolis and gum arabic to control postharvest anthracnose and maintain quality of papaya during storage”. International Journal of Agriculture and Biology 16 (2014): 1117-1122.
- Anu Aravind A., et al. “Phytochemical analysis and antioxidant potential of the leaves of Garcinia travancorica Bedd”. Natural product research 30 (2014): 232-236.
- Attalla KM., et al. “Antibacterial activities of bee venom, propolis, and royal jelly produced by three honey bee, Apis mellifera L, hybrids reared in the same environmental conditions”. Annals of Agricultural Sciences Moshtohor 45.2 (2007): 895-902.
- Badr A.N., et al. “Ochratoxin a occurrence on Egyptian wheat during seasons (2009-2014)”. Asian Journal of Scientific Research 10 (2007): 178-185.
- Badr AN., et al. “Anti-mycotic and anti mycotoxigenic properties of Egyptian dill”. Journal of Applied Sciences 17: 184-195.
- Balouiri., et al. “Methods for in vitro evaluating antimicrobial activity: A review”. Journal of Pharmaceutical Analysis 6 (2007): 71-79.
- Chacko CM., et al. “Antimicrobial evaluation of jams made from indigenous fruit peels”. International Journal of Advanced Research 2.1 (2014): 202-207.
- Demissie N. “A Review of Aflatoxin: Occurrence, Prevention, and Gaps in Both Food and Feed Safety”. Journal of Food Science and Technology 1.3 (2018).
- Esper RH., et al. “Potential of essential oils for protection of grains contaminated by aflatoxin produced by Aspergillus flavus”. Frontiers in microbiology 5 (2014): 269.
- Falca o SI., et al. “Phenolic characterization of Northeast Portuguese propolis: usual and unusual compounds”. Analytical and Bioanalytical Chemistry 396 (2014): 887–897.
- Fawole OA., et al. “Antibacterial, antioxidant and tyrosinase-inhibition activities of pomegranate fruit peel methanolic extract”. BMC Complementary and Alternative Medicine 12:200 Fumonisin B (2012).
- “Sometraditionalherbalmedicines, some mycotoxins, naphthaleneandstyrene”. IARC monographsontheevaluation of carcinogenic risks to humans.
- Groth CG., et al. “Sirolimus (rapamycin)-based Therapy In Human Renal Transplantation: Similar Efficacy and Different Toxicity Compared with Cyclosporine1, 2”. Transplantation 67 (1999): 1036-1042.
- Guo C., et al. “The antioxidant activities of peels, pulp and seed fractions of common fruit as determined by FRAP assay”. Nutrition Research 23.13 (2003): 1719-1726.
- Hasnaoui N., et al. “Valorization of pomegranate peel from 12 cultivars: dietary fibre composition, antioxidant capacity and functional properties”. Food Chemistry 160 (2014): 196-203.
- Hwang ES., et al. “Effects ofextraction and processing methods on antioxidant compound contents and radical scavenging activities of laver (Porphyra tenera)”. Preventive Nutrition and Food Science 19 (2014): 40-48.
- IARC. “Monographs on the evaluation of carcinogenic risks to humans: chemical agents and related occupations.A review of human carcinogens”. Lyon, France: International Agency for Research on Cancer 100 (2012): 224-248.
- Ismaiel AA., et al. “Mycotoxins: Producing Fungi and Mechanisms of Phytotoxicity”. Agriculture 5 (2015): 492-537.
- Kapadiya DB., et al. “Spices and Herbs as a Source of Natural Antioxidants for Food”. International Journal of Current Microbiology and Applied Sciences 5.7 (2016): 280-288.
- Keno T., et al. “Major Biotic Maize Production Stresses In Ethiopia And Their Management Through Host Resistance”. African Journal Of Agricultural Research13 (2018): 1042-1052.
- Labyad N., et al. “Phytochemical antioxidant and antimicrobial study of Libyan propolis ethanolic extract”. International Journal of Herbal Medicine 4.5 (2016): 01-04.
- Li Y., et al. “Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract”. Food Chemistry 96 (2016): 254-260.
- Lu Y., et al. “Ionic liquid-based microwave-assisted extraction of phenolic alkaloids from the medicinal plant Nelumbo nucifera Gaertn”. Journal of chromatography42.6 (2008).
- Mc Clave., et al. “Statistical for Business and Economics. Max Well Macmillan International Edn”. Publisher: Dellen Publishing Company USA 272-295 (1996).
- Mohdaly., et al. “Phenolicextractfrompropolis and beepollen: composition,antioxidant and antibacterial activities”. Journal of Food Biochemistry 39 (1996): 538–547.
- Narbona E., et al. “Volatile composition of functional ‘a la Piedra’turro´n with propolis”. International Journal of Food Science and Technology 45 (1996): 569-577.
- Noah BA., et al. “Anti-mycotic and Anti-mycotoxigenic properties of Egyptian dill”. Journal of Applied Sciences 17 (2017): 184-195.
- Nooh A., et al. “Mycotoxin and toxigenic fungi occurrence in Egyptian maize”. International Journal of Advanced Research 2 (2014): 521-532.
- Orak H., et al. “Effects of hot air and freeze drying methods on antioxidant activity, colour and some nutritional characteristics of strawberry tree (Arbutus unedo L) fruit”. Food science and technology international = Ciencia y tecnologia de los alimentos internacional18 (2012): 391-402.
- Ozsoy N., et al. “Antioxidant activity of Smilax excelsa L. leaf extracts”. Food Chemistry 110 (2008): 571–583.
- Pagliarulo C., et al. “Inhibitory effect of pomegranate (Punica granatum L.) Polyphenol extracts on the bacterial growth and survival of clinical isolates of pathogenic Staphylococcus aureus and Escherichia coli”. Food Chemistry 190 (2016): 824–831.
- Picman AK., et al. “Antifungal activities of sunflower terpenoids”. Biochemical Systematics and Ecology 18 (1996): 325–328.
- Ramadan MF., et al. “Functional characteristics, nutritional value and industrial applications of Madhuca longifolia seeds: an overview”. Journal of food science and technology53 (2016): 2149-57.
- Rather RA., et al. “Screening of peel extracts as antioxidants, anticancer agents and antimicrobials”. Advances in Bioresearch 1.1(2010): 29–33.
- Re R., et al. “Antioxidant activity applying an improved ABTS radical cation decolorization assay”. Free Radical Biology and Medicine 26.19 (1999): 1231-1237.
- Sakanaka S., et al. “Preparation and antioxidant properties of extracts of Japanese persimmon leaf tea (kakinoha-cha)”. Food Chemistry 89 (2005): 569-575.
- Salman. “HDAntibacterial Activity of Propolis Extracted in Three DifferentSolvents and in Three Different pH values on somePathogenic Bacteria”. International Journal of Pharm Tech Research 9.10 (2005): 258-266.
- Sforcin JM. “Propolis: Is there a potential for the development of new drugs”. Journal of Ethnopharmacology 133 (2016): 253–260.
- Shehata MG., et al. “Oil-bioactive Films as an Antifungal Application to Save Post-harvest Food Crops”. ARRB 16.4 (2017): 1-16.
- Turkmen N., et al. “The effect of cooking methods on total phenolics and antioxidant activity of selected green vegetables”. Food Chemistry 93 (2014): 713–718.
- Vodnar DC., et al. “Identification of the bioactive compounds and antioxidant, antimutagenic and antimicrobial activities of thermally processed agro-industrial waste”. Food Chemistry 231 (2005): 131 - 140.
- Waliyar F., et al. “Institutionalizing Mycotoxin Testing in Africa”. In: Leslie Education (2008): 367-375.
- Waliyar F., et al. “Pre-and post-harvest management of aflatoxin contamination in peanuts. Mycotoxins: detection methods, management, public health and agricultural trade”. CABI, Wallingford, UK (2018): 209-218.
- Zahra P., et al. “Application of Modern Agricultural Practices to Reduce Aflatoxins Exposure in Developing Countries”. Nutrition and Food Toxicology 2.3 (2007): 346-347.
Citation:
Abdelazim Sayed Abdelazim., et al. “Applications of Some Natural Active Extracts for Cereal Storage Improvement”. Nutrition
and Food Toxicology 3.2 (2018): 604-617.
Copyright: © 2018 Abdelazim Sayed Abdelazim., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.




































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.