Case Report
Volume 2 Issue 3 - 2018
A Rare Case of Asymptomatic Cavernous Sinus Thrombosis in Neuromyelitis Optica Spectrum Disorder
1Department of Neurology, Kuala Lumpur Hospital, Malaysia
2Department of Radiology, Hospital Kuala Lumpur, Malaysia
2Department of Radiology, Hospital Kuala Lumpur, Malaysia
*Corresponding Author: Joyce Pauline Joseph, Consultant Neurologist, Department of Neurology, Hospital Kuala Lumpur, Kuala Lumpur, Malaysia.
Received: August 28, 2018; Published: September 06, 2018
Abstract
Neuromyelitis optica spectrum disorder (NMOSD) is an inflammatory disease of the central nervous system that includes transverse myelitis, optic neuritis, acute brainstem syndrome, area postrema syndrome, symptomatic narcolepsy or acute diencephalic clinical syndrome with NMOSD-typical diencephalic lesions on magnetic resonance imaging, and symptomatic cerebral syndrome with NMOSD-typical brain lesions. NMOSD has been associated with autoimmune conditions and rare cases of venous thrombosis. A 60-year-old woman who presented with recurrent optic neuritis and transverse myelitis was diagnosed with aquaporin 4 antibody–positive NMOSD. She presented with blurring of vision and was found to have right Optic Neuritis and incidental left cavernous sinus throbombosis. She was treated with methylprednisolone and improvement in symptoms were noted. To our knowledge, this is the first documented case of cavernous sinus thrombosis in NMOSD.
Key Words: Neuromyelitis Optica Spectrum Disorder; Cavernous Sinus Syndrome; Venous Thrombosis; Autoimmune; Demyelinating
Abbreviations: Neuromyelitis Optica Syndrome Disorder (NMOSD); Magnetic Resonance Imaging (MRI) If there are any abbreviations in the article they have to be mentioned
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an inflammatory disease of the central nervous system. [1] The International Panel for NMO Diagnosis revised previous diagnostic criteria for neuromyelitis optica with more stringent criteria to create the unifying term NMOSD. It is classically characterized by involvement of the optic nerve, spinal cord and brain. NMOSD has been associated with various autoimmune diseases such as rheumatoid arthritis, Sjögren’s syndrome, systemic lupus erythematous, thyroid disease, myasthenia gravis, ulcerative colitis, antiphospholipid syndrome and autoantibodies without diseases. [2] The pathophysiology and mechanism of these associations have not been recognized. However, there have been no reports of cavernous sinus thrombosis to date. Cavernous sinus syndrome consists of multiple cranial nerve involvement, Horner’s syndrome, ophthalmoplegia, orbital congestion, proptosis, trigeminal sensory loss and sympathetic impairment. [3,4]. However, patients can be asymptomatic and may not fulfil the criteria. We report a rare case of carvernous sinus thrombosis in a patient with NMOSD.
Case Report
A 60 year-old lady, a restaurant manager by profession, had recurrent episodes of optic neuritis and transverse myelitis. Investigations revealed she was aquaporin 4 antibody positive with magnetic resonance imaging (MRI) suggestive of demyelinating disease. She was given azathioprine but could not tolerate the drug due to neutropenia and liver impairment. There have been treatment delays due to an earlier misdiagnosis of multiple sclerosis and an unfortunate situation with a renal abscess that required the prolonged use of antibiotics. Her mobility has been restricted due to recurrent transverse myelitis. Interferon therapy was initiated in September 2013 and continued until May 2014. There was one episode each of urinary tract infection and right perinephric abscess in May of 2014 and August of 2014, respectively.
Azathioprine was given from March 2014 until May 2016. In March 2016 she presented with blurring of vision and double vision. Clinical examination revealed a relative afferent pupillary defect over the right eye. Vision over the right eye revealed no perception to light and the left eye acuity was 6/15. Ishihara’s colour vision test revealed deficiencies in both eyes with scores of 0/15 on the right and 1/15 on the left. Colour vision could not be assessed on the right side but fundoscopy revealed pale optic discs bilaterally; eye movements were full and there was no evidence of ophthalmoplegia. There was no speech or swallowing difficulty, tone was normal, power in the upper limbs was 5/5 bilaterally and reflexes were normal. Lower examination revealed normal tone, 4/5 power bilaterally and brisk reflexes. Plantar reflex showed Babinski’s sign.
She was treated with intravenous methylprednisolone for acute optic neuritis. A computerized tomography scan revealed and incidental fullness of the left cavernous sinus region. MRI confirmed bulky cavernous sinus in the left and enhancing intracanalicular right optic nerves slightly compressed by dural thickening-Figure 1 (A and B). Intraorbital parts of the optic nerve were normal. Both optic nerves showed nerve sheath enhancement more on the right side, which also indicates prechiasmatic involvement. A repeat MRI upon completion of steroid treatment revealed resolved bulkiness of cavernous sinus region. Figure 2(A and B) There were no clinical or radiological features suggestive of central nervous system tumours such as meningioma, neurofibroma, nasopharyngeal carcinoma or metastatic lesions. No evidence of carotico-cavernous fistula or cavernous sinus aneurysm was seen. The patient did not have clinical features suggestive of connective tissue disease and there was no other evidence of infective cause. She declined lumbar puncture and cerebral venous study for further evaluation
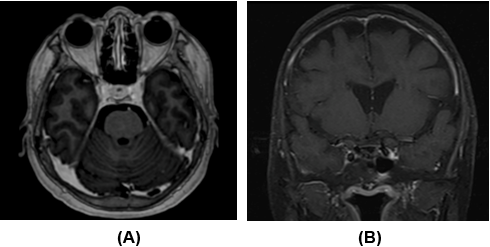
Figure 1: Initial pretreatment (25/3/2016) T1-post-gadolinium
magnetic resonance imaging in axial (A) and coronal (B) planes
demonstrate a bulky and thickened wall of the left cavernous sinus.
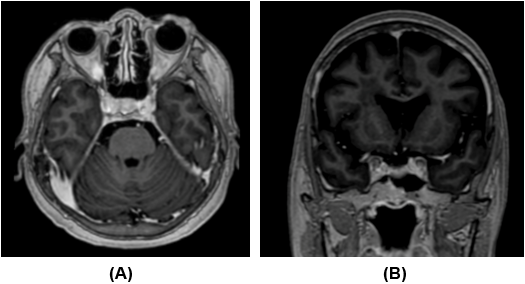
Figure 2: Post-treatment (26/4/16) T1-post gadolinium magnetic
resonance imaging in axial (A) and coronal (B) planes; the left
cavernous sinus wall thickening has been reduced in size.
Discussion
NMOSD is a rare immune-mediated disease which has evolved in recent years in terms of diagnosis, clinical description and disease associations. Several other conditions have been associated with NMOSD. There has been no association with cavernous sinus syndrome or thrombosis reported to date. Our patient presented with visual symptoms and she was diagnosed and treated for optic neuritis as that would be the commonest diagnosis for her presentation. She was given intravenous steroids. She did not present with typical features such as impaired extraocular muscle movement, chemosis or congestion of the eyes, Horner’s syndrome or trigeminal nerve involvement for venous thrombosis in the cavernous region. In view of the patient receiving immunosuppresants in the past, she was investigated for chronic infective central nervous lesions such as tuberculosis but the investigations did not reveal any positive results.
Neuroimaging done after a course of methylprednisolone showed no evidence of cavernous sinus thrombosis. The authors feel that the pathophysiology behind cavernous sinus thrombosis in this patient could be due to an inflammatory process associated with her background autoimmune condition. A histopathological diagnosis would have been a valuable assessment because such a presentation has never been reported and studied. Our patient did not consent to a biopsy for confirmation of the diagnosis.
We feel that rare associated conditions will need to be considered in patients with demyelinating disorders such as NMOSD so that all visual symptoms are not diagnosed as optic neuritis. That the cavernous sinus lesion improved after steroids is an indication that her cavernous sinus abnormalities could be immune mediated. In their study of 151 cases of cavernous sinus syndrome, Keane., et al. [5] found self-limited inflammation is the third most common cause after tumour and trauma. The authors feel more case studies and research are required to confirm this association.
Conclusion
Cavernous Sinus thrombosis is rare in Neuromyelitis Optica Syndrome Disorder and can be an asymptomatic manifestation
Conflict of interest: None
References
- Wingerchuk DM., et al. “International consensus diagnostic criteria for neuromyelitis optica spectrum disorders”. Neurology 85.2 (2015): 177-189.
- Yanqiang W., et al. “Neuromyelitis optica spectrum disorders with autoimmune diseases”. Journal of Multiple Sclerosis 3 (2016): 167.
- Lee JH., et al. “Cavernous sinus syndrome: clinical features and differential diagnosis with MR imaging”. American Journal of Roentgenology 181.2 (2003): 583-590.
- Bone I and Hadley DM. “Syndromes of the orbital fissure, cavernous sinus, cerebello- pontine angle, and skull base”. Journal of Neurology, Neurosurgery, and Psychiatry 76.S3 (2005): iii29-iii38.
- Keane JR. “Cavernous sinus syndrome. Analysis of 151 cases”. Archives of Neurology 53.10 (1996): 967-971.
Citation:
Joyce Pauline Joseph., et al. “A Rare Case of Asymptomatic Cavernous Sinus Thrombosis in Neuromyelitis Optica Spectrum
Disorder”. Medical Research and Clinical Case Reports 2.3 (2018): 232-235.
Copyright: © 2018 Joyce Pauline Joseph., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.



































 Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.
Scientia Ricerca is licensed and content of this site is available under a Creative Commons Attribution 4.0 International License.